Pharmaceutical Company

BUSINESS PLAN
PAIN AWAY LTD.
1117 High St.
Poughkeepsie, NY 13495
The company described in this plan has moved beyond the initial start-up phase and is now seeking investors to finance its growth. Much of the plan, therefore, is geared toward persuading, explaining, and reassuring potential investors that the company (which produces a therapeutic, topical pain cream), is well-managed and stable. The in-depth analysis of the company's competitors is an outstanding feature of this plan, as is its market research.
- EXECUTIVE SUMMARY/OVERVIEW
- MARKET
- COMPETITION
- MARKETING
- PRODUCTION
- PROPERTY & FACILITIES
- PATENTS & TRADEMARKS
- RESEARCH & DEVELOPMENT
- GOVERNMENT REGULATIONS
- INSURANCE AND TAXES
- CORPORATE STRUCTURE
- RISK FACTORS
- RETURN ON INVESTMENT AND EXIT
- ANALYSIS OF OPERATIONS & PROJECTIONS
- FINANCIAL STATEMENTS
EXECUTIVE SUMMARY/OVERVIEW
Type of Business
Non-prescription drug wholesalers; US SIC Code - 2834 Pharmaceutical Preparations.
Company Summary
Pain Away Ltd. is a going concern, a Delaware corporation formed in January 1995 to manufacture and sell its premier launch product Pain Away, a topical pain remedy using FDA-approved homeopathic ingredients developed for the simple purpose of relieving pain. The company was formed by its parent S-corporation, Peale, Inc. in order to market products nationally and internationally. Peale, Inc. was formed in February 1994 to complete the development of the launch product. The formation of the company was a significant step in a 9-year process of refining and testing a homeopathic formula first used by company founder and CEO Robert Peale to alleviate his pain from carpal tunnel syndrome. The R&D phase of this product began when Mr. Peale purchased the original formula, did a thorough study of homeopathy, and refined the formula to its present marketable state. From the beginning of R&D, Mr. Peale worked within FDA guidelines in order to secure FDA registration. Then, in February 1994, the company was formed to finally manufacture and sell the product. Starting with only a handful of customers, including some professionals, chiropractors, physical therapists, etc., only 19 months of operation have yielded 12,000 individual customers with an 80% reorder rate. The current customer base now includes medical doctors from different specialties, sports trainers, and athletes, both professional and amateur. The company expects to show a profit in 1996 and estimates that it will be very profitable in 3 years.
Management
Mr. Peale is 49 years old and has a 25-year history in sales, sales management, and marketing for a tool distribution company. His deep study of homeopathic medicines started in 1985 and included studies in nutritional supplements. Mr. Peale has been invited to sit on a newly-formed FDA committee addressing the growing national interest in natural medicines.
Curtis Company president, Ms. Alana has 25 years of experience in retail and direct sales. She has been a senior sales director and sales trainer for Beautiful You Cosmetics, has owned and operated a retail sporting goods store, and has managed a 15 person, $1 million department for a major chain retailer. She also has some banking experience.
Vice-president of marketing, Ryan Lemon has 32 years of experience as production manager, buyer, sales manager, and marketing manager. He was director of marketing for Pilgrim Health and was responsible for their first launch into New Jersey which led to their first $18MM in sales (in 3 years). He has a BS degree in textile engineering and has also done independent marketing consulting.
Product and Competition
The R&D mission was to develop a greaseless, odorless, topical cream which was measurably more effective at relieving pain than any other OTC (over the counter) topical product. This mission has been accomplished. The company has collected anecdotal, testimonial, and uncontrolled medical study evidence that Pain Away is more effective than the leading topical analgesics such as Arthritin and others. The product's effectiveness in relieving pain is its most powerful benefit, besides the added benefits of it being greaseless and odorless. What distinguishes Pain Away from any other topical analgesic in this still-growing $402.1MM market is its advanced homeopathic formula - a refined blend of 11 FDA approved pure and natural ingredients. The typical OTC topical analgesic works to either block the sensation of pain or distract perception of deep pain by "counterirritating" another localized area near the pain. Pain Away's formula is different. Pain Away treats pain at its source. It stimulates improved circulation in the micro-capillary system in the ligaments and tendons, where most pain is felt. Pain-relief from Pain Away is the result of the body's own self-healing. It also can be applied several times a day because it is odorless and greaseless. The US pain management market ($15.2 billion by 1997) is a mature market with intense, established competition ("The Market for Pain Management Products in the US - Introduction, Drugs, Devices, Trends, and Market Structure," in FIND-SVP). With future pharmaceutical market growth dependent upon new and innovative product additions, Pain Away is entering the field at the right time. The company will distinguish itself and its market position by dedication to the development of only natural-ingredient products. Since its unique formula of ingredients already has FDA approval, the company aims to penetrate the OTC pharmaceutical market, where new products traditionally find success. Here Pain Away will compete with topical as well as internal analgesics, including aspirin, acetaminophin and ibuprofen. An estimated 4,000 people a year die from aspirin overdose. A condition known as "analgesic neuropathy" can result from extended or inappropriate use of analgesics. Medical studies linking heavy usage to health problems have affected aspirin, acetaminophin, and ibuprofen. Pain Away can be marketed as a substitute for (reducing overdose risk with internal analgesics), or as a supplement to (using Pain Away can reduce needed dosage of internal analgesics) internal analgesics when used for certain pain relief. Furthermore, Pain Away is not contraindicated for use with any other medication. This broad-based appeal is built upon the reliability of Pain Away's effectiveness in relieving pain, inflammation, and spasm associated with arthritis, bursitis, sciatic spasm, neck/back pain, tendonitis, tennis elbow, tension headache, achilles tendonitis, and carpal tunnel syndrome.
A second product, a natural anti-inflammatory nutritional support system formula known as "Pain Away Plus," will soon be marketed as a companion product to Pain Away. This multistaged formula is a combination of trace minerals, herbs, and a natural cartilage-derived substance. The company has long-term plans to develop more health-related products.
Funds Requested
Company principals have invested all available personal assets into the product development and operations to date. The need for capital is in the context of the readiness of the product for mass marketing. Management is seeking a $1,500,000 equity investment in exchange for a suggested 30% ownership of the company. All terms of financing are negotiable in order to meet the financial requirements of the investor.
Use of Proceeds
Advertising & promotion campaign - $1,200,000 (see below); Market research - $300,000. The company anticipates the need for follow-on financing after 24 months of business.

| Advertising & Promotion | Projected Cost |
| Magazines | $330,000 |
| Radio | $200,000 |
| Shows & Conventions | $140,000 |
| TV | $400,000 |
| Retail Shops | $70,000 |
| Sample - POP Display | $60,000 |
| Total | $1,200,000 |
Financial History

| From 5/94-12/31/95 | |
| Sales | 543,633 |
| Net Income | (226,600) |
| Assets | 56,987 |
| Liabilities | 224,253 |
| NetWorth | (167,266) |
Sales were first made in 5/94 under Peale Inc. ($143,881). As sales expanded nationally, Pain Away Ltd was formed in January 1995. All sales since then have been under Pain Away Ltd.
Financial Projections
| 1996 | 1997 | 1998 | 1999 | 2000 | |
| Sales | 3,000,000 | 8,000,000 | 18,000,000 | 32,000,000 | 50,000,000 |
| Net Inc. | 360,000 | 2,160,000 | 4,860,000 | 8,640,000 | 13,500,000 |
With capital request accomodated, the company believes that Pain Away will jump in sales starting in 1996.
Exit
The company will attempt a public offering based on year 2000 earnings. If there is no public market and no prospect for a public market in the near future, then the company will offer to buy back the stock owned by the venture capitalist. A predetermined price could be set ahead of time, if desired by the venture capitalist.
PRODUCT
The product effectiveness, evidenced largely through anecdotal evidence, personal testimonials, and repeat sales, has formed the basis for the future growth of the company. Together with a second, complementary product (nearly ready for market), the launch product will be aggressively mass marketed as a pain management system for the next five to ten years. Past and current sales have been to end-users, health professionals, and to some retail chains. The company and product are now poised for first stage expansion. Over 30 target wholesale markets have been identified. While the company uses its marketing strategy to enter these wholesale markets, simultaneous efforts will be made to develop research protocols. Management is confident that the anecdotal evidence and personal testimonials will be strengthened by controlled studies, designed to test the effectiveness of the product and demonstrate the physiological healing activity stimulated by the formula. With scientific credibility, the product will not only build its position in the $150 million homeopathic product category but will also strengthen its transition into the formidable mainstream topical analgesic category.
Future research is planned, based upon inquiry, in order to adapt the formula for animal use (Pain Away currently being tested on thoroughbred horses).
At the end of five years, the company intends to have at least one additional health product and should be able to go public off its revenues. The long-term goal for the company is to become an entrepreneurial leader in the development of natural products for various segments of the health care market. The company plans to capture enough share of the topical analgesic market to become either a viable joint venture partner or an acquisition candidate.
Uniqueness
The product formula and delivery system are proprietary. The formula is uniquely advanced and is nearly immediately effective in relieving pain. Homeopathy and immunization have much in common, namely the principle of similars, which states that whatever a substance causes in a large dose, it can stimulate an immune response to defend against it in a small dose. It works by the principles of stimulation to the body's own self-healing mechanism and by the scientific balancing of its natural active ingredients through a dilution process called micro-dosing. Micro-dosing has given homeopathy its 200-year history of safety with no known side effects or toxicity. This self-healing process is in contrast to the majority of commercially successful topical analgesics, which contain counter-irritants, including the newer capsaicin-based products. These ingredients cause a superficial inflammation on the skin which masks pain by deadening the sensation of pain in the epidermal nerve endings only, or by distracting from the perception of pain by irritating an area near the pain source. The Pain Away formula has been developed with precision and balance and is a product that is effective and safe for use on all skin types. Pain Away's eleven active ingredients stimulate improved circulation in the micro-capillary system to ligaments and tendons, where most pain is felt. Pain relief is the result of the body's self-healing.
The manufacturing is sub-contracted out to a highly respected FDA-licensed manufacturer of homeopathic products.
An important unique feature of Pain Away which distinguishes it from other homeopathic remedies is that Pain Away is a topical treatment and is not a systemic treatment. As such, it requires little knowledge to use and is conducive to cross-merchandising in the mainstream analgesic category. Furthermore, since Pain Away is a formula of ingredients, it provides a broad spectrum of effects as compared to single remedies.
The personal commitment of the founder to relieve his own pain also adds a unique value to the story of this product - a story which can enhance marketability - to anyone who is in pain or anyone who knows someone in pain.
Although Pain Away is an homeopathic product, the company will position itself as a natural ingredients company - not necessarily homeopathic. All the company principals plan to engage both septics and advocates of complementary medicine by applying rigorous scientific standards equally across the board, for both conventional and unconventional treatments. Contacts have already been made with the National Institute of Health regarding future research.
Product Description
The product is a specialty consumer good carrying a suggested retail price of $19.95 for a 3.7 oz. jar (1.9 oz. jar also available at $12.95). The jar is designed with a medical appearance. The jar is easy to ship in multiples, is easy to stack on a shelf, is aesthetically pleasing, and has an easy-to-handle screw cap. The actual cream is greaseless, easy and pleasant to apply, and is odorless. Pain Away has, to date, largely been sold directly to end-users, and wholesale to retailers, distributors, and catalogues. The markets have supported the suggested retail price, which was arrived at by surveying market research supporting the $19.95 price along with the perceived value of the product compared to similar products at about the same price. This price also yielded a gross profit of $3.75 per jar and allowed for 100% markup from wholesale.
The eleven ingredients are readily available through top-quality labs which control for purity and authenticity. The cream is compatible with any medication being taken. The product carries a money-back 30-day guarantee.
Purchasers of the Product
Preliminary studies done by independent treatment professionals (no control group used) have shown that Pain Away has been effective for relieving the pain, inflammation, and spasm associated with arthritis, bursitis, sciatic spasm, neck and back pain, tendonitis, tennis elbow, tension headache, achilles tendonitis, and carpal tunnel syndrome. Anyone suffering these ailments, treating these ailments, or caring about anyone suffering these ailments is a potential purchaser of the product. A New Jersey hockey team uses Pain Away prior to workouts, competition, and for pain relief. The head trainer for the team says, "There's no product better for contusion of the quadriceps." He has reported shorter recovery times as a result of using Pain Away. Reports from athletes are that using Pain Away before and after workouts yields less cramping, fatigue, and soreness.
Top purchasers of TPR to date:
| Customer | Dollar Sales | Unit Sales |
| Mall Booth Marketing | $11,000/month | 800/month |
| Direct Selling-Retail | $16,000/month | 1200/month |
MARKET
The total market for OTC internal and topical analgesics is estimated at $3.6 billion for 1995 and is projected to be $4 billion by 1997. With over 400 brands saturating this mature market, growth is still occurring through new products and product innovations. Driving this growth are:
- increasing use of pain management products for the over-50 population segment, whose numbers are increasing
- increasing awareness that pain does not have to be tolerated and can be treated
- price increases
Body/Muscle Pain Market
The market is dominated by internal analgesics:
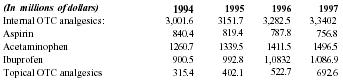
| (In millions of dollars) | 1994 | 1995 | 1996 | 1997 |
| Internal OTC analgesics: | 3,001.6 | 3151.7 | 3,282.5 | 3,340.2 |
| Aspirin | 840.4 | 819.4 | 787.8 | 756.8 |
| Acetaminophen | 1260.7 | 1339.5 | 1411.5 | 1496.5 |
| Ibuprofen | 900.5 | 992.8 | 1,083.2 | 1/086.9 |
| Topical OTC analgesics | 315.4 | 402.1 | 522.7 | 692.6 |
Pain Away is a new product to this sizable OTC pain-relief market. It will enter this large arena riding on its effectiveness and coming from the new and growing alternative health care market segment. As a new OTC product, Pain Away has such a broad-based appeal that it will be sold to a large portion of the total OTC pain-relief market (both internal & topical), estimated to be 84% of all US adults and growing as the baby boom population ages and concerns regarding age-related ailments, such as arthritis, increase. Of this 84%, about 25% alone use pain-relief products for body/muscle pain for which Pain Away is especially suited. Just this one type of ailment offers a substantial market potential:
| Population size | %need product | Frequency Use | = | Mkt. Potential |
| 161.3 Million X | 40.3 Million X | 156x/year | = | 6.3 Billion uses |
If only 40.3 million Americans (25% of 84% of adults) use an OTC pain-relief product three times a week for body/muscle pain alone, then the market potential is 6.3 billion uses of a pain-relieving product per year. Past use of Pain Away has indicated that a minimum of 3 applications per week would use about one 3.7 oz. jar per month. A conservative yearly estimate would be 10 jars per year, with consistent use. In order to reach a five-year sales goal of $50 million (6.7 MM jars), 667 MM consistent purchasers (10 jars/yr.) are needed. Product history has indicated a consistent 80% reorder rate, so at this rate, 833,000 original purchasers are required. This figure is 2.07% of just this one market segment. The company is very confident that it can capture 2.07% of this market segment within five years, especially considering that the roughly 40 million Americans who exercise on a regular basis, and who are aging, are included in this segment. Anecdotal reports from athletes who use Pain Away are that it can prevent injuries by "warming up" vulnerable muscles and joints prior to a workout. The product has wide applicability within this segment. The table below shows the percentage of the body/muscle pain market segment required to meet the next 5 years of sales projections.

| Year | Sales Goal | Initial Purchasers | Needed % of Market |
| (in millions) | (with 80% reorder rate) | ||
| 1996 | $3 | 50,000 | .12% |
| 1997 | $8 | 133,000 | .33% |
| 1998 | $18 | 300,000 | .74% |
| 1999 | $32 | 533,000 | 1.32% |
| 2000 | $50 | 833,000 | 2.07% |
These numbers are based upon a wholesale price of $7.50 per jar and a usage rate of 10 jars/year with a segment population of 40.3 million potential purchasers.
The prescription pain relief market is a distinct market which Pain Away will not attempt to penetrate. Pain Away can, however, compete directly with nearly all pain-relief products because of its unique identity of being both a substitute and a supplement to ail competing products. This uniqueness fits a projected market shift from internal to topical analgesic use as the population ages, and derives from 2 factors: 1) Use of Pain Away can reduce the needed dosage of any pain-relieving medication and 2) Pain Away is already part of a rapidly growing segment (25%-30%/year) of consumers who use alternative health care because of a disenchantment with OTC drugs and a concern about side effects with adverse reactions. Use of Pain Away can reduce needed dosage of other pain-relieving medications. As stated earlier, Pain Away's effectiveness is based upon the homeopathic principle of microdosing. While it promotes self-healing by stimulating blood flow to micro-capillaries, it remains safe for all skin types and with use of any other medication. Anecdotal evidence (from hospitals, some doctors, and occupational rehab center) has indicated that use of Pain Away alone has yielded positive results and use of Pain Away, along with other treatments, has seemed to accelerate recovery. As always, this kind of evidence will be scientifically studied. The salient point is that Pain Away can be a substitute and/or a supplement in pain management, and thereby reduce needed dosages of other medications.
Alternative Health Care Segment
Homeopathy, being an established (officially recognized by UK National Health Service) and significant alternative mode of treatment, is gaining increasing acceptance in mainstream American health care. The National Institute of Health has even awarded grant money for research in alternative treatments, including homeopathy. Drug retailers report that homeopathy may be the fastest-growing category in the trade class of drug chains. Since homeopathy is gaining acceptance as an alternative treatment, the market segments which are already embracing these alternatives will continue to be targeted in the company's initial expansion. These segments include people ages 25-elderly, who seek improved quality in life, and whose lifestyle values involve "newness." This segment includes most of the "baby-boomer" population, estimated at over 75 million. The market of alternative health care seekers is characterized by patients who can and will pay for their own care. As much as 70% of alternative medical treatments are still paid for by patients themselves rather than insurers. This kind of purchasing indicates a willingness to try an alternative product and continue purchasing based upon perceived value of the product's effectiveness. Company management has been encouraged by the consistent 80% reorder rate and knows sales will be sustained once initial purchases are made. The alternative health care market is of respectable proportion. According to the New England Journal of Medicine (1/28/93), 34% of Americans spend $13 billion/year on alternative treatments such as chiropractic, acupuncture, massage, and homeopathy. Pain Away is already marketed to all of these treatment specialties so it will reach the spectrum of alternative treatment. This 34% of Americans are familiar with the term "homeopathic," so there's a consumer predisposition to being further educated about homeopathy as a value-added natural ingredient alternative.
The company will build an early market position on the alternative health-care market and will join the growth of the homeopathic segment as it moves from the fringes to the mainstream of the OTC pharmaceutical market.
Alternative Market Potential:
| Population size | % need product | Frequency Use | = | Mkt. Potential |
| 262 Million × | 89.1 M (34%) × | 24x/Yr. | = | 2,000 Billion |
If only about one third of Americans use an an alternative pain-reliever just twice per month, then the market potential is 2 trillion uses of an alternative pain-relieving product per year. Market indicators are that both the number of users and the frequency of use will increase as the population ages. The use rate of 2 times per month converts to 2 jars of Pain Away per year with consistent use. Again, in order to reach the 6.7 million jar sales goal ($50 MM), at the re-order rate of 80%, Pain Away would have to make 4.2 million initial sales in order to sustain 3.3 million consistent purchasers. This size customer base comprises 4.71% of the growing alternative health care market. The company believes that this sales goal is attainable within the next five years. The table below shows the percentage of the alternative health care market segment required to meet projected sales.
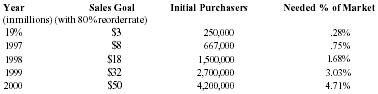
| Alternative Health Care Market | |||
| Year | Sales Goal | Initial Purchasers | Needed % of Market |
| (in millions) | (with 80% reorder rate) | ||
| 1996 | $3 | 250,000 | .28% |
| 1997 | $8 | 667,000 | .75% |
| 1998 | $18 | 1,500,000 | 1.68% |
| 1999 | $32 | 2,700,000 | 3.03% |
| 2000 | $50 | 4,200,000 | 4.71% |
These numbers are based upon a wholesale price of $7.50 per jar and a usage rate of 2 jars/year with a segment population of 89.1 million potential purchasers.
Narrowing the Market Focus 2X
The market potential for pain relief products is huge. By narrowing the focus to product category sales, the potential becomes more exact. Pain Away's product category is within the topical analgesic market, estimated at $402.1 MM annually with a projected $522.7 MM market in 1996 (30% growth) and $692.6 in 1997 (32.5% growth). Starting with $522.7 as the base market volume, and with 30% growth per year for the next 5 years, Pain Away would have to capture 3.33% of the year 2000 market volume to make its sales goal of $50MM. Management believes that these goals are attainable.
The table below shows what percentage of the topical analgesic market will meet Pain Away's sales projections.
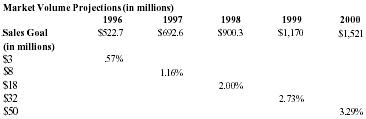
| Topical Analgesic Market | |||||
| Market Volume Projections (in millions) | |||||
| 1996 | 1997 | 1998 | 1999 | 2000 | |
| Sales Goal | $522.7 | $692.6 | $900.3 | $1,170 | $1,521 |
| (in millions) | |||||
| $3 | .57% | ||||
| $8 | 1.16% | ||||
| $18 | 2.00% | ||||
| $32 | 2.73% | ||||
| $50 | 3.29% | ||||
The focus can be narrowed further to the homeopathic product category, which is growing at a rapid rate at this time. The dollar volume of this segment is estimated at present to be between $150 million and $215 million and expected to grow at a rate of 25% to 30% a year. Some market-trackers say that retail sales haven't grown enough to support the existing number of homeopathic manufacturers and that a shakeout will consolidate sales in the hands of fewer manufacturers. The forseeable trend, however, is progressive growth from the fringes to mainstream markets, and at a rapid rate. The table below again shows percentages of this dollar volume required to meet sales projections.
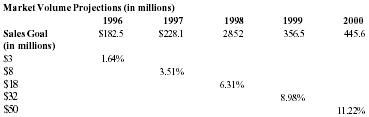
| Homeopathic Products | |||||
| Market Volume Projections (in millions) | |||||
| 1996 | 1997 | 1998 | 1999 | 2000 | |
| Sales Goal | $182.5 | $228.1 | 285.2 | 356.5 | 445.6 |
| (in millions) | |||||
| $3 | 1.64% | ||||
| $8 | 3.51% | ||||
| $18 | 6.31% | ||||
| $32 | 8.98% | ||||
| $50 | 11.22% | ||||
These numbers are based upon a 1996 volume mid-point between the projected volume range of $150 MM and $215 MM. Growth rate is 25% a year. At first glance these percentages may seem daunting. However, the manufacturers supplying this niche are relatively few in number and therefore hold significant market shares A new player can get a reasonable market share with the right product and marketing plan. The mainstream merchandising of homeopathic products started in the early '90's and has been tested as a lucrative direction. Company management is very confident that Pain Away will gain enough share points to capitalize on the rapid growth of this product category. Pain Away will not remain in the homeopathic niche. Its effectiveness will make it competitive with mainstream topical angalgesics.
International Markets
The company will also develop an international market. A 10,000-unit order has already been received from a distribution company in Hungary and is awaiting final approval from the Hungarian State Department of Pharmacy. A small order was also sent to well-known sports figure in Spain. Discussions are underway for this individual to start large-scale distribution. The homeopathy market in the UK is estimated at 18M pounds and in Germany at 120M pounds, so European marketing could be strengthened by the homeopathic identity alone. In Germany, an independent division of the German Federal Health Agency publishes monographs on the safety and efficacy of herbal medicines. The company believes that Pain Away would fare excellently under such review and will carefully research and plan when and how to reach such markets.
COMPETITION
There are many companies competing for shares of the 3.6 billion dollar OTC analgesic market. The major players are the internal analgesic manufacturers:

| Company | Product | Analgesic Sales-$ | Mkt. % |
| Reynolds | Aspernol | 1.2B | 34% |
| Pharmacorp | Aspiril | 612 MM | 17% |
| American Pharmacy | Anaprin | 180 MM | 5% |
| Oxford Co. | Maraprin | 180 MM | 5% |
| Jones-Smythe Benton | Aspirin | 144 MM | 4% |
The balance of the OTC internal analgesic market is held by private label companies and "others." The major strengths of this level of competition are obvious in comparison to Pain Away's present market position. The major players have:
- a manufacturing cost advantage,
- sophisticated market knowledge and access,
- established sales capability,
- strong R&D capacity,
- and of course, brand name loyalty.
An important competitive strength of Pain Away is that it is topical - pain relief is accomplished without risk of overdose and consequent risk of serious side effects. This competitive strength derives from a previously noted shift in the market from internal to topical analgesic use. This shift in consumer preference, along with Pain Away's effectiveness, can position the product as a substitute/supplement among these large competitors. Management is ever mindful that mainstream pharmaceutical companies are watchful of the homeopathic market and will act accordingly should market share be lost to homeopathic remedies. Becoming a viable acquisition candidate to any one of its major competitors is a realistic goal. Pain Away management is committed to quality product development and is also open to strategic alliances which would enhance its market capability.
The competition in the topical analgesic market is head-to-head. The top competitors are:

| Company | Product | Ingredients | Sales | %Share |
| Pepper Co. | Pepperub | Menthol | 60.3 MM | 15% |
| Athens | Vapol | Menthol | 47.9 MM | 11.9% |
| Lucia | Menthol Plus | Menthol | 35.8MM | 8.9% |
| Skin Care Corp. | Zanprin | Capsaicin | 44.2 MM | 8.7% |
| Bioderm | Aspratin | Salycin | 18.8 MM | 5% |
| Capcreme | Capsaicin | NA | ||
| Men-Thol Co. | Menthoflex | Menthol | NA | |
| Capthol | Capsaicin | Menthol | NA | |
| Bianco-Picard | Salicreme | Methylsalicilate | 86 MM | |
| Synergy | Lyptum | Eucalyptus | NA |
The basis for the competitive analysis is Pain Away's most competitive feature:
- It doesn't have any of the aforementioned advantages held by the major, well-known players in this market - yet.
- It doesn't have widespread brand name recognition - yet.
- It doesn't have appreciable market share in topical analgesics, alternative health, or homeopathy - yet.
- It does have a unique formula of safe and effective ingredients which none of the above products have.
All topical analgesics contain counter-irritants, including camphor, menthol, methyl salicylate, eucalyptus, wintergreen, and even the popular capsaicin. These ingredients, even when blended, act primarily to cause a superficial inflammation on the skin. This inflammation serves to hide the pain by deadening pain receptors in the skin.
What distinguishes Pain Away from all of the above products is that the eleven active homeopathic ingredients stimulate the blood flow in the body's micro-capillaries and act synergistically with the body tissue. This stimulates the body's own self-healing. Pain is treated at its source. Company management believes that the unique effectiveness of Pain Away will give it competitive clout. The issue then becomes how to compete.
Although Pepperub (Pepper) and Vapol (Athens) enjoy the largest market share, they are vulnerable to new product introductions. Menthol Plus (Lucia) held the top position in this category last year until Pepperub was re-packaged and relaunched with line extensions. That relaunch along with a relaunch of Zanprin boosted sales of both brands and put Pepperub back on top. Pepperub, Vapol, and Mentholplus are all menthol-based products. Zanprin is a capsaicin-based product and has boosted usage of its relatively new ingredient. Other relatively new capsaicin products are Capcreme (Bioderm) and Capthol (Men-Thol Co.).
Company management believes that Pain Away is generally more effective than Pepperub and Vapol. However, these venerated brand names, large advertising budgets, and consumer loyalty are formidable competitive advantages. Pain Away will focus on other competitors in order to gain a market position.
The key competitors are Menthol Plus, made by Lucia and Zanprin, made by Skin Care Corp.. Menthol Plus is a menthol-based product which Pain Away has encountered head to head in the sports market. Menthol Plus has a retail price advantage in the mass market, selling for $4 for a 2 oz. tube. This price difference is of little concern because Pain Away will promote itself as a high value product. The topical analgesic, alternative health, and homeopathic markets all support pricing based on perceived product value. Menthol Plus' manufacturer has reduced the advertising budget for this product (about $2 million) recognizing from a 21% decrease in 1994 sales that the product has matured. The company plans to acquire other brands (no topical analgesics) and extend its other lines in order to generate sales growth. The company sells another topical analgesic which is doing well in sales but has not reached the same position as Menthol Plus. Pain Away will monitor the life cycle of Menthol Plus and move to gain any market share it might lose.
Zanprin, made by Skin Care Corp., is gaining market share because Zanprin (.025%) and Zanprin- X (.075%) are capsaicin-based products. Capsaicin, derived from cayenne peppers, has created a new segment in the market and is very popular. Other companies are making capsaic in products but Skin Care Corp. attracted market attention by relaunching Zanprin as an OTC consumer product. It had been marketed for seven years to physicians and kept behind the counter, carrying the credibility of a prescription product. In early 1995, the product was re-packaged for shelf space and supported by TV ads. Despite commanding premium prices ($19.95/2oz of Zanprin-X), the product has done dramatically well.
Skin Care Corp. claims that Zanprin is the "only brand with physician endorsement and specific clinical support." This is a credible claim, cultivated for seven years, and obviously contributing to sales of the product.
Skin Care claims to be the first in the industry to develop their highly purified version of capsaicin for a pharmaceutical base. Zanprin distinguishes itself by promoting controlled clinical studies which have supported its effectiveness. Skin Care claims that such clinical trials don't apply to other, less pure, capsaicin formulas. This scientific feature enhances product credibility among physicians and pharmacists.
The management of Pain Away Ltd. recognizes the effective marketing strategy used by Skin Care because it is similar to their own strategy. Advertising and promotion expense is critical. With proper capitalization, Pain Away can compete because the Pain Away homeopathic formula is unique and effective. Many capsaicin users, including Zanprin users, have complained about the burning sensation caused by capsaicin. Pain Away will stand up to any topical analgesic on the market and do very well with comfort, safety, and effectiveness. The company needs to get this message out. The seven-year product life of Zanprin, supported by unique and heavy TV advertising, gives Zanprin quite an edge. Zanprin is now a "new" growth product and Pain Away can grow behind it, by comparing ingredients and effectiveness at every turn. Pain Away is also in the same price range as Zanprin, doing slightly better with $19.95 for a 3.7 oz. jar or $12.95 for a 1.9 oz. jar.
Zanprin is not the "only brand with physician endorsement and specific clinical support." Pain Away has been cultivating health professional support since the R&D phase. The product is heavily endorsed, and more medical support is developing. Many of Pain Away's sales to date have been to health professionals. Regarding clinical support, Skin Care's success with this strategy underscores the strategic importance of Pain Away's plans for controlled clinical studies.
Speaking of "highly purified" formulas, Pain Away can compete strongly with any formula on the market, especially capsaicin-based. The company wants to discuss purity of ingredients and formula and will do so in all promotional efforts.
The remainder of the products listed in the top competitor list have of course the same advantages that any established company with significant market share has. Beyond these immediate competitive advantages, Pain Away can compete, again, on the ingredient effectiveness basis.
Aspratin, an odorless rub which contains Salycin, sold well when it was introduced in 1992. It held third place among topical analgesics at the end of 1993. It has since been surpassed by capsaicin-based Zanprin. Bioderm developed Capcreme and lowered its price when Zanprin was relaunched.
Capthol was recently developed by the long-established Men-Thol Co. and is a capsaicin-menthol blend designed to compensate for the sometimes delayed pain relief when using capsaicin alone.
Salicreme is a methylsalicylate product which has shown flat growth and has lost market share.
Lyptum was a rapid-growth product in 1990-1991 but has since lost market share. Besides the well-established brands like Pepperub, the products which are gaining in this market are the capsaicin-based. This product category is known to be affected by product innovation and development. With proper support, Pain Away will take a respectable market share.
Homeopathic Competition
The competition takes place in the drug chain arena. Homeopathy may well be the fastest-growing category in the trade class of drug chains (20% of all homeopathic product sales). Among the growing number of drug chains which are giving shelf space to homeopathic products are: Walgreens, Medicine Shoppes International, Thrifty Payless, Eckerd Corp., Edgehill Drugs, Genovese and FEDCO, a California supermarket chain. Research published in the Journal of Clinical Pharmacy and Therapeutics states that 27% of US pharmacists consider homeopathic medicines helpful while only 18% consider them useless. The crossover of homeopathy from health food stores, where sales are still strong, to mass markets is gaining momentum.
As mentioned earlier, there are relatively few companies supplying homeopathic products to the mass market. There are five major producers/distributors of homeopathic products.

| Company | Product |
| Health System Products | Full line of products |
| Homeopathic Co. | Full line under brand name Organa |
| Life-Right Corp. | Full line |
| Del Sol Inc. | Full line |
| Scandinavian Co. | Full line |
| Bio Health | Full line |
Health System, Homeopathic Co., and Life-Right pioneered the distribution of homeopathic products to chain drug stores in the early 1990's and are now market leaders, although more companies are entering this lucrative market. Health System Products now has about 40% market share. Homeopathic Co. and Del Sol are aggressively developing the crossover into mass marketing with line development and heavy TV advertising.
All the topical analgesics listed above are arnica-based, with few other ingredients. Arnica Montana is the premier homeopathic medicine for the treatment of shock and trauma to the muscle. These formulas come the closest to Pain Away's because they contain some of the essential homeopathic pain-reducing ingredients. Pain Away's formula, however, blends more ingredients than any other homeopathic topical analgesic on the market. This more inclusive formula gives the product wider applicability. Price-wise, Pain Away is more expensive than most of the competing homeopathic products, where prices are in the $5-$10 range for 2oz.-4oz. sizes. But, this is a value-priced market, so price is not a critical variable. Since Pain Away is very competitive on an ingredient/effectiveness basis, the critical factor is having the resources to promote the product.
Future Competition
As has been noted, the topical analgesic category, including natural ingredient, is rapidly influenced by new clinical studies and product innovations. There are three main sources of new competition:
- New ingredients and/or new innovations of existing ingredients. Examples are new products which employ the medicinal benefits of ammonium compounds. These products are designed to provide pain relief without the objectionable training room smells, burning sensations and stinging of abraded skin that are often caused by the majority of topical analgesics that contain menthol, methyl salicylate or capsaicin as active ingredients. Pain Away's formula has solved this sensation problem and is a less "high-tech" product, for which consumers are showing a preference.
- Companies currently in this market who could increase market share and become major players. Pain Away Ltd. is in this category.
- Chain drug companies may produce their own private label homeopathic products and corral a significant share of this growing market - much as they did in the non-homeopathic analgesic market. This scenario is more likely to happen as homeopathic companies expand the sales volume in this market and there are share points to be taken away by private labeling.
Pain Away Ltd. can be very competitive with the right promotional support.
MARKETING
Marketing Strategies
Increase market share by reducing market share of competitors. This strategy will capitalize on the market development to date and capture a share of markets held by existing pain-relieving topical applications. The key benefit is that conventional pain-relievers mask pain while Pain Away stimulates the body's own healing ability to directly battle an ailment. Another benefit is that homeopathic remedies have no known side effects while many pain-relievers, especially those ingested, have side effects. Neither will Pain Away interfere with any medication. This strategy requires extensive advertising in mainstream media, including infomercial, QVC (Pain Away already under review), 60 second commercial, cable TV, interactive TV, direct mail, independent sales reps, POP displays, and educational inserts/newsletters. One objective of planned controlled studies on the effectiveness of Pain Away is to use scientific evidence to help bridge the narrowing gap between natural and conventional medicine. Product studies will support this marketing strategy. In this context, the company will pursue preliminary inquiries from a favored vendor to use Pain Away in the workplace to study any reduction of lost work time and/or medical costs precipitated by repetitive stress injuries.
Expand a growing new market for alternative health care by positioning to lead this growing market. This strategy involves specialty catalogues (placed in 5 currently), placement on retail shelves of health food stores, educational product inserts/newsletters, media appearances discussing product, and independent sales reps. This strategy addresses the 89.1 million users of alternative health care.
The company has already been approached by two large Multi-Level Marketing companies. This strategy would involve creating private labels for a large customer. Of utmost consideration with this strategy is product identity and how this channel of distribution would affect it. This channel of distribution usually requires more price mark-up than the product would tolerate.
The company will create its own "competition" by developing private labels and/or separate companies to market to different niches.
Keep capital outlay to a minimum by licensing/franchising Pain Away to a brand-name company. This strategy would add value to the product in the form of brand name loyalty, manufacturing strength, and a strong sales/service force already in place. The company envisions its role in this type of strategic alliance as conducting scientific studies to increase the credibility of TPR and in developing new products. This strategy remains an option which could preclude other strategies under mutually acceptable terms.
Building on an initial order from a health product distribution company in Hungary, Pain Away Ltd. will penetrate the European market by targeting England and Germany, where homeopathy is an accepted form of treatment. This strategy would be developed only after a US market position was established.
Marketing Plan
The company is moving from start-up stage into its first growth stage. Market strategy to date can be succinctly described as selling "one jar at a time." Direct personal selling has been the mainstay in sales growth. This strategy has targeted any end-user willing to try the product. These early customers were reached through health care professionals and direct selling through state/county fairs, shopping mall space, health food store chains, and most recently lifestyle catalogues. As the company moves away from direct selling, a strategy which proved to be an excellent market test, into mass-marketing, identified market segments are being matched with appropriate distribution channels. The plan now is to expand and concentrate more on helping the consumer develop product preference by heavy advertising of the brand name, the benefits of the product, the ease of use, and the guarantee. Company expectations are that all advertising will be enhanced by results of controlled studies of product effectiveness.
The company intends to expand regionally, based on existing markets and consumer profiles (e.g., households from the South are likely heavy users of analgesics). The national market will only be tested by placement in catalogues with a distribution of 200 million. As regional sales grow and as the product gains recognition, then a national marketing strategy will take shape. Company management have begun discussions with a major marketing communications agency (Fortune 500 client list) who themselves approached Pain Away. The marketing and sales outline is as follows.
Marketing Function
Research
- A complete review and analysis of the topical analgesic market.
- Utilization of Triad Groups conducted with the professional community and general consumers. Purpose is to identify professional and consumer preferences.
- Based on research, create a product identity.
- From product identity, establish professional and consumer strategic directions, which would affect product design, packaging, advertising, consumer promotion, and product publicity.
- Test both professional and consumer strategic direction via two more Triad Groups.
- Develop launch marketing plan with all elements and budget for both professional and consumer.
- Actual implementation of the plan to include product design changes, packaging, advertising, consumer promotion, display, and product publicity.
Sales Function
Retail
Utilize a sales organization enabling direct-call coverage on the top 25 customers, which generally account for 80% of retail sales, and broker-managed coverage for the remainder. Launch plan would include a national sales meeting and all necessary materials.
Professional
Concentrate on the pharmacist community via co-op direct mail. Pharmacist recommendation at the purchase counter does affect sales.
PRODUCTION
The production process takes place in a standard homeopathic laboratory where raw materials are blended. There are no significant health or safety risks involved. Production orders are processed by purchase order for finished product. Some raw materials are usually on hand but more are ordered against purchase order requirements. Jars are ordered from a separate manufacturer and sent to the homeopathic laboratory to be filled, packaged, and shipped to Pain Away Ltd., where fulfillment is done.
The homeopathic laboratory has the capacity to fill all projected orders. As orders increase, Pain Away management will consider using a fulfillment service and more drop-shipping to wholesale customers. Cost of goods is estimated at 18% of gross sales. This figure has been consistent throughout production to date and is based on the complete production cycle.
There is no backlog.
Production Characteristics
The production process does not require any specialized or proprietary machinery. The critical factors in the production process are the highest quality of raw materials and the incubation process, which assures a stable finished product. Water is added to a base of vegetable/plant emollients. The eleven active ingredients are then mixed into the emulsion, which incubates for about 48 hours in large vats, while monitored for any fungal invasion. The finished product is then lab-tested for potency, which is done by lot number (the company gets lot samples). Filling is currently done by gravity-feed. The manufacturer might advance to computerized filling. One batch is 500 gallons. Lead time from order to packaged product is 4 weeks. Only a skilled and experienced manufacturer can produce the formula. Even other homeopathic manufacturers not familiar with a cream-based product would have difficulty with the production process. General topical analgesic manufacturers would need to become familiar with the raw materials and the production process in order to blend Pain Away's eleven active and ten inert ingredients. The company currently has one back-up manufacturer, which has never been used.
Labor Force and Employees
The company administrative staff consists of 5 people (recently reduced by 3) including the 3 officers. The two employees are paid an hourly wage. The staff are not unionized and there is no expectation of such. The labor supply in the region is more than sufficient to meet all future staffing needs. The sales force is comprised of independent agents who are paid on commission.
Suppliers

| Supplier | Volume | Product |
| Herbal Laboratories | 35,000 jars | all raw materials |
| Portland, Oregon | jars & caps | |
| labels | ||
| packaging | ||
| shipping boxes |
Currently, the laboratory procures all production materials. There are no shortages of key components, and multiple sources are available.
Subcontractors
All production is sub contracted out. Only fulfillment and shipping are done in-house. The company has formed a strong working relationship with Herbal Laboratories, which is the key subcontractor. Although management has selected a back-up manufacturer, the existing relationship with Herbal Labs has been more than satisfactory, so no change is foreseen. Other subcontractors supplying jars, labels, and boxes are used based upon price and service and can be replaced.
Equipment
Standard office equipment is used for administrative functions. All production equipment at Herbal Laboratories is new and there is nothing that would cause production to be stopped for any appreciable time.
PROPERTY AND FACILITIES
The company facility is a single-story 1,950 square foot, cement block structure on about a two-acre cleared lot that is leased in one-year increments. The facility is located in northern Dutchess County, NY. All necessary commercial and industrial infrastructure is in place. The facility is easily accessible from major thoroughfares. The general area has been and is recovering from the closing of 2 large industrial facilities, so there's been anoticeable decline in property values. There is, however, a regional effort to re-direct the area to rely more upon small and entrepreneurial business. Management plans to purchase the building in order to add an appreciable fixed asset and to reduce expenses. The structure is easily expandable, so the company will not have to move during its critical growth stage.
PATENTS AND TRADEMARKS
Active homeopathics are not patentable. Management is exploring establishing a trademark and a formula patent.
RESEARCH AND DEVELOPMENT
The three principals have invested collectively $100,000, which has been capitalized. Plans for the immediate future include forming a research alliance with a university, hospital, or research group in order to develop a protocol for applying the "rigorous scientific standards" against which the effectiveness of Pain Away can be proven. Management has projected R&D expenses at $ 30,000 for the next 12-month period. These expenditures are intended for controlled studies proving effectiveness, and for continuation of developing applications for animals. Management is sales-marketing oriented and does not want to develop only a research lab. Any R&D will be designed to enhance sales and profits. Company management is currently investigating an SBIR grant.
GOVERNMENT REGULATIONS
There are no particular federal, state or local laws/regulations that affect the conduct of business. The manufacturer meets OSHA requirements, as does the Pain Away administrative facility. The FDA regulates homeopathy as an OTC non-prescription medicine. Pain Away's ingredients are in total compliance with FDA standards. Mr. Peale cultivated a working relationship with FDA representatives during the initial research and wisely intends to sustain such.
INSURANCE AND TAXES
Product liability insurance is underwritten. A buy-sell agreement among officers exists but is not yet backed by insurance. Key employee insurance is also yet to be written.
All taxes are current. The company pays standard payroll, Social Security, and corporate taxes. The product is sales tax exempt in many states.
CORPORATE STRUCTURE
Company principals first formed an S-corporation under the name Peale Inc. The realization of the likelihood of international sales prompted management to form Pain Away Ltd. as the operational company. Peale Inc. serves a limited partnership which was formed to attract investors. Both companies are run by the same management team. All R&D is done through Peale Inc. There is comingling of funds. This proposal seeks financing for Pain Away Ltd. Return on the investment will derive from the sale of the product Pain Away itself and any other products which the company sells.
Pain Away Ltd. is a member of the Homeopathic Manufacturers Association. The officers were invited to participate in an annual meeting of the newly formed FDA committee on natural medicines. This committee works on the bases for regulations, compliance, and claims for the natural ingredient industry, covering vitamins, herbs, and homeopathy.
Management subscribes to the following publications:
- Homeopathy Today
- Natural Foods Merchandiser
- American Health
- Prevention Magazine
- Let's Live
- New England Journal of Medicine letter
Directors and Officers
A board of directors will be developed in the near future. There is interest from the medical, nutritional, and professional sports communities, as well as from a local bank. Officers are:
Robert Peale - CEO
Alana Curtis - President
Ryan Lemon - Vice-President, Marketing
Profit and loss responsibilities are shared by the officers.
The officers are primary key employees (backgrounds in executive summary). Other key employees include:
Key Employees
Leslie Ottaviani - bookkeeper and office manager - known by management for 5 years and described as "a dedicated innovator with a true grasp for details." She has experience supervising 20 employees in the accounting department of Worldwide Airlines and has worked as an independent bookkeeper for several companies in Hudson Valley, NY.
Julia Allen - administrative assistant - known by management for 6 years and described as "having people and problem-solving skills and works incredibly well under pressure." Her background includes sales in a successful business which included business consulting.
Remuneration
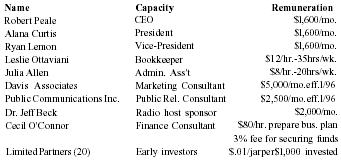
| Name | Capacity | Remuneration |
| Robert Peale | CEO | $1,600/mo. |
| Alana Curtis | President | $1,600/mo. |
| Ryan Lemon | Vice-President | $1,600/mo. |
| Leslie Ottaviani | Bookkeeper | $12/hr.-35hrs/wk. |
| Julia Allen | Admin. Ass't | $8/hr.-20hrs/wk. |
| Davis Associates | Marketing Consultant | $5,000/mo.eff. 1/96 |
| Public Communications Inc. | Public Rel. Consultant | $2,500/mo.eff. 1/96 |
| Dr. Jeff Beck | Radio host sponsor | $2,000/mo. |
| Cecil O'Connor | Finance Consultant | $80/hr. prepare bus. plan |
| 3% fee for securing funds | ||
| Limited Partners (20) | Early investors | $.01/jarper $1,000 invested |
Accountant and Banker
| Jonathan Wainwright | Accountant | no retained\fee for service only |
| Arnold Lee | Banker | no remuneration |
All other fees paid on an ad hoc basis. Different attorneys have been used on an ad hoc basis (finance closing fees will be paid by the company).
Principal Shareholders
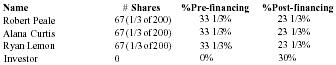
| Name | #Shares | %Pre-financing | %Post-financing |
| Robert Peale | 67 (1/3 of 200) | 33 1/3% | 23 1/3% |
| Alana Curtis | 67 (1/3 of 200) | 33 1/3% | 23 1/3% |
| Ryan Lemon | 67 (1/3 of 200) | 33 1/3% | 23 1/3% |
| Investor | 0 | 0% | 30% |
FINANCING
Proposed Financing
Management is willing to negotiate any structure which suits the investor. The company is seeking an equity investor. Management will provide a seat on the company's board of directors. Ongoing reports of key ratios, profit-loss statements, balance sheets, and annual audits would be provided to the investor. It is management's intent that the investor will enjoy returns on investment in excess of that of alternative investments, as a privately held company, while providing investor liquidity of his investment by taking the company public at its earliest opportunity.
Capital Structure
The existing capital structure includes a $50,000 unsecured line of credit with Poughkeepsie National Savings Bank. This line of credit was just brought to maturity in 1/96 for a 30-day period, at which time the line was renewed. If the current financing proposal is accomodated, then the line of credit can be increased.
Additional financing to date has derived from the sale of limited partnerships offering $.01 per 3.7 oz. jar royalty for every $1,000 invested. Each limited partner has been given the right to convert his/her capital investment into common stock when the company goes public, or, to receive back his/her original capital investment when the company goes public. Total amount of financing raised through the limited partnership is $100,000.
As mentioned earlier, officers have collectively invested about $100,000 in the company, mostly through the R&D phase. Officers' "sweat equity" is immeasurable.
Use of Proceeds
As stated in the executive summary: Advertising & promotion campaign - $1,200,000 (see below); Market research - $300,000. The company anticipates the need for follow-on financing after 24 months of business.

| Advertising & Promotion | Projected Cost |
| Magazines | $330,000 |
| Radio | $200,000 |
| Shows & Conventions | $140,000 |
| TV | $400,000 |
| Retail Shops | $70,000 |
| Sample-POP Display | $60,000 |
| Total | $1,200,000 |
Management intends to preserve cash flow by factoring much of the receivables. With the current lead time of 4 weeks, however, some capital may be used to increase merchandising inventory in order to fulfill initial large orders. It is hoped that any follow-on financing can and will be debt financing, serviced by cash flow.
The following table sets forth the capitalization of Pain Away Ltd. as of 12/31/95 and as adjusted to reflect the proposed sale of common stock.
Dilution

| Stockholders Equity | Before sale Actual | After sale As Adjusted |
| Common Stock, no par value, | (167,268) | 1,332,732 |
| 200 shares authorized; | ||
| 0 outstanding | ||
| Additional Paid-in Capital | 1,500,000 | |
| Accumulated Earnings (deficit) | (167,268) | (167,268) |
| Total Stockholders' Equity | (334,536) | 1,165,464 |
Dilution: The net tangible book value of the company as of 12/31/95 was minus $1,673 per share. Without taking into account any other changes in such net tangible book value after 12/31/96, other than to give effect to the sale of 60 shares (proposed 30% equity share) hereby, the pro forma net tangible book value of the company on 12/31/95 will be $5,827 per share, representing an immediate dilution of $13,597 per share to new investors.

| Price per share to Investor | 19,424 |
| Net tangible book value before the sale | (1,673) |
| Increase attributable to new investor | 7,500 |
| Pro forma net tangible book value after the purchase | 5,827 |
| Dilution to new investor | $13,597 |
Management recognizes that this proposed financing implies a large premium value on the existing equity and so will negotiate any other conditions which would induce the investor to make the investment.
At the time of the company's IPO, limited partners who opt for common stock will receive their shares from the officers' share of owned stock. The negotiated ownership held by the investor will not be further diluted.
Investor Involvement
Management seeks a close working relationship with the investor. The investor will be given one seat on the board of directors. Management would solicit consultations (for a fee) on financial matters, or any other area of investor expertise (e.g., planning, management development), but voting power is not an option. Fees will also be paid for any future financing and/or profitable business connections arranged by the investor.
RISK FACTORS
Limited Operating History
Even though management feels that the company is at first-stage expansion, it is definitely still an early-stage company. Two obvious risks inherent in early-stage companies are undercapitalization and poor liquidity. Management has capitalized the business operations to date well enough to have developed the product and identified penetrable market segments. The current proposed financing will provide enough capital to handle the anticipated growth.
Limited Resources
Management believes that it has the resources to continue at the present pace of business. An anticipated increase in sales through advertising media such as QVC , regional/national catalogues, retail outlets, and some European distribution can be financed by factoring. These "bootstrapping" approaches have sustained the company to date and will accommodate slow growth. Management believes, however, that more rapid expansion is desirable in order to penetrate its identified market segments. More rapid expansion requires more resources.
Limited Management Experience
All officers have successful backgrounds in marketing. Additional experience in manufacturing/distribution has been gained in the past nine years of product development. Management has consistently shown a willingness to leverage themselves with accomplished professional consulting relationships. The company culture is one which reinforces sharing of expertise with mutual benefit to all concerned.
Market Uncertainties
Any consumer product business is subject to the changing preferences of the marketplace. As presented in the marketing section of this proposal, the target markets are showing substantial growth, which limits uncertainty. There is currently a growing consumer preference for homeopathic topical remedies. More uncertainty is evident when considering competition, but can be made tolerable by on-going research and analysis.
Production Uncertainties
The only uncertainty at present is whether or not the lead time (4 weeks) from purchase order to finished product can consistently be reduced. This uncertainty is of material concern as sales increase. Herbal Laboratories is a sound company with a promising long-term future and has always been customer-friendly, so no more serious uncertainties exist at present. Management believes that vertical integration of manufacturing is feasible in the long-term but is not practical in the near-term.
Liquidation
In the event that liquidation becomes necessary, management believes that the most value could be realized from the sale of the product formula itself. The formula is not patented, so valuation remains uncertain. However, the sales history, along with the testimonials attesting to the effectiveness of this "ready-made" product, should determine value. Office equipment would yield limited value, and unless the company building was purchased prior to liquidation, no value would be realized. Management believes that the company can and will generate increasing value in the near future, evidenced by increasing sales.
Dependence on Key Management
At present, CEO Robert Peale is considered the primary key manager/officer. His knowledge of the product ingredients, his history of public appearances promoting the product, his increasing recognition by the health community as an expert in natural medicine, and his charisma as a business professional highlight his key role. Managerially, the other officers are thoroughly competent and could manage the company and market its products without Mr. Peale. At this critical early stage, however, the product needs an identity and a market position before the loss of any key managers could be overcome. Once the premier product is securely launched and the product line is expanded, the loss of any officer could be absorbed by continued proper management of the company. Management believes that such a development is not far off, once the company is properly capitalized. Until such time, key person life insurance will be purchased.
What Could Go Wrong?
Upgraded advertising campaigns could not lead to any substantial increase in sales. This problem can be avoided by using experienced advertising/marketing consultants who have familiarity with the targeted markets. Furthermore, properly designed test runs on any advertising campaign would provide objective indicators of expected returns. Capital investment in advertising should be gradual and progressively based upon certain expected levels of return.
Stronger competition could capitalize on and stall Pain Away's early success by replicating the product and its marketing strategy. This problem can be solved in two ways: First, with proper capitalization, Pain Away can make an entry into targeted markets rapidly and with enough strength to grab market share. Keeping market share can be easier than getting it. This market requires extensive advertising. Increasing market share could mean an increasing advertising budget. An increasing advertising budget can easily reduce profit margin, so strategic planning is required. The second way to solve the competition problem is in the formula itself. Management will seek to patent the formula. The nature of the homeopathic ingredients is likely to inhibit any mainstream non-homeopathic company from replicating the product. Acquisition of a homeopathic company would make more sense. Narrowing the competition, then, to other homeopathic companies gives Pain Away more of a fighting chance, since its formula is more sophisticated and user-friendly than any homeopathic topical analgesic on the market.
Governmental controls could conceivably impede sales. This problem is unlikely because the ingredients are already FDA-approved. Furthermore, management's participation in the FDA committee to develop regulatory standards for the natural medicine field would provide early warnings of any such prohibitory controls.
The company could be controlled by non-investor stockholders. This problem is not likely to develop because the management team would hold a majority. Management is dedicated to the principles of increasing value and profits and is confident that its efforts will be in concert with those of the investor.
RETURN ON INVESTMENT ANDEXIT
Public Offering
Management plans for an IPO in 5-7 years. The investor's shares would be sold to provide the targeted return on investment. Should there be no public market, then a buy back would occur.
Buyback
Management will negotiate a buy back formula with the investor and will target milestones in planning for this possibility. Management aims for returning 6 times the original investment in five years.
ANALYSIS OF OPERATIONS AND PROJECTIONS
The business has not shown a profit since sales activity began in May 1994. This lack of profit is not unusual for an early-stage company. Losses were incurred in the start-up phase, where the objective was to get consumers to try the product. Gross profit margins have remained stable, however. Management focus was targeted on getting professionals and consumers to try the product in order to collect anecdotal evidence and testimonials of its effectiveness. Not enough focus was on asset management, as evidenced by a low return on assets ratio (p.32). Now that the product has gotten some recognition, especially in professional circles, the focus will shift toward mass marketing. Management intends to improve inventory management by using factoring of receivables in conjunction with JIT inventory control. As sales volume increases, drop-shipping from plant to wholesale customer, will also be arranged.
FINANCIAL STATEMENTS
Balance Sheet
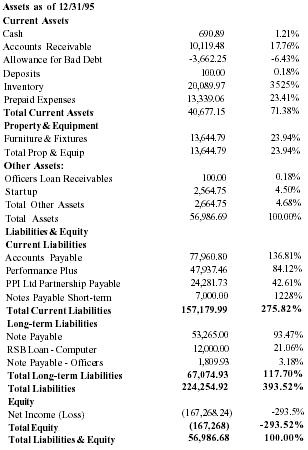
| Assets as of 12/31/95 | ||
| Current Assets | ||
| Cash | 690.89 | 1.21% |
| Accounts Receivable | 10,119.48 | 17.76% |
| Allowance for Bad Debt | −3,662.25 | −6.43% |
| Deposits | 100.00 | 0.18% |
| Inventory | 20,089.97 | 35.25% |
| Prepaid Expenses | 13,339.06 | 23.41% |
| Total Current Assets | 40,677.15 | 71.38% |
| Property & Equipment | ||
| Furniture & Fixtures | 13,644.79 | 23.94% |
| Total Prop & Equip | 13,644.79 | 23.94% |
| Other Assets: | ||
| Officers Loan Receivables | 100.00 | 0.18% |
| Startup | 2,564.75 | 4.50% |
| Total Other Assets | 2,664.75 | 4.68% |
| Total Assets | 56,986.69 | 100.00% |
|
Liabilities & Equity
Current Liabilities |
||
| Accounts Payable | 77,960.80 | 136.81% |
| Performance Plus | 47,937.46 | 84.12% |
| PPI Ltd Partnership Payable | 24,281.73 | 42.61% |
| Notes Payable Short-term | 7,000.00 | 12.28% |
| Total Current Liabilities | 157,179.99 | 275.82% |
| Long-term Liabilities | ||
| Note Payable | 53,265.00 | 93.47% |
| RSB Loan - Computer | 12,000.00 | 21.06% |
| Note Payable - Officers | 1,809.93 | 3.18% |
| Total Long-term Liabilities | 67,074.93 | 117.70% |
| Total Liabilities | 224,254.92 | 393.52% |
| Equity | ||
| Net Income (Loss) | (167,268.24) | −293.5% |
| Total Equity | (167,268) | −293.52% |
| Total Liabilities & Equity | 56,986.68 | 100.00% |
Monthly Income Statements 1995
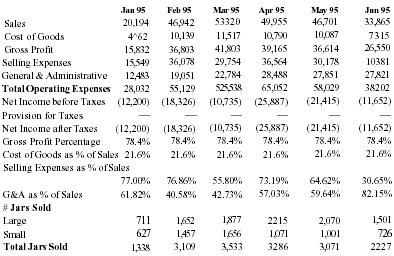
| Jan 95 | Feb 95 | Mar 95 | Apr 95 | May 95 | Jun 95 | |
| Sales | 20,194 | 46,942 | 53,320 | 49,955 | 46,701 | 33,865 |
| Cost of Goods | 4,362 | 10,139 | 11,517 | 10,790 | 10,087 | 7,315 |
| Gross Profit | 15,832 | 36,803 | 41,803 | 39,165 | 36,614 | 26,550 |
| Selling Expenses | 15,549 | 36,078 | 29,754 | 36,564 | 30,178 | 10,381 |
| General & Administrative | 12,483 | 19,051 | 22,784 | 28,488 | 27,851 | 27,821 |
| Total Operating Expenses | 28,032 | 55,129 | 525,538 | 65,052 | 58,029 | 38,202 |
| Net Income before Taxes | (12,200) | (18,326) | (10,735) | (25,887) | (21,415) | (11,652) |
| Provision for Taxes | — | — | — | — | — | — |
| Net Income after Taxes | (12,200) | (18,326) | (10,735) | (25,887) | (21,415) | (11,652) |
| Gross Profit Percentage | 78.4% | 78.4% | 78.4% | 78.4% | 78.4% | 78.4% |
| Cost of Goods as % of Sales | 21.6% | 21.6% | 21.6% | 21.6% | 21.6% | 21.6% |
| Selling Expenses as % of Sales | 77.00% | 76.86% | 55.80% | 73.19% | 64.62% | 30.65% |
| G&A as % of Sales | 61.82% | 40.58% | 42.73% | 57.03% | 59.64% | 82.15% |
| # Jars Sold | ||||||
| Large | 711 | 1,652 | 1,877 | 2,215 | 2,070 | 1,501 |
| Small | 627 | 1,457 | 1,656 | 1,071 | 1,001 | 726 |
| Total Jars Sold | 1,338 | 3,109 | 3,533 | 3,286 | 3,071 | 2,227 |
Income Statement - 12/31/95
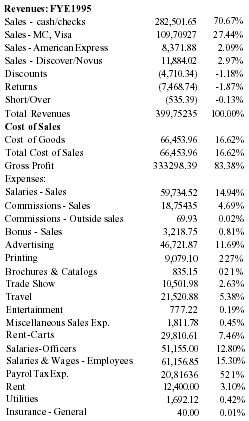
| Revenues: FYE 1995 | ||
| Sales - cash/checks | 282,501.65 | 70.67% |
| Sales - MC, Visa | 109,709.27 | 27.44% |
| Sales - American Express | 8,371.88 | 2.09% |
| Sales - Discover/Novus | 11,884.02 | 2.97% |
| Discounts | (4,710.34) | −1.18% |
| Returns | (7,468.74) | −1.87% |
| Short/Over | (535.39) | −0.13% |
| Total Revenues | 399,752.35 | 100.00% |
| Cost of Sales | ||
| Cost of Goods | 66,453.96 | 16.62% |
| Total Cost of Sales | 66,453.96 | 16.62% |
| Gross Profit | 333,298.39 | 83.38% |
| Expenses: | ||
| Salaries - Sales | 59,734.52 | 14.94% |
| Commissions - Sales | 18,754.35 | 4.69% |
| Commissions - Outside sales | 69.93 | 0.02% |
| Bonus - Sales | 3,218.75 | 0.81% |
| Advertising | 46,721.87 | 11.69% |
| Printing | 9,079.10 | 2.27% |
| Brochures & Catalogs | 835.15 | 0.21% |
| Trade Show | 10,501.98 | 2.63% |
| Travel | 21,520.88 | 5.38% |
| Entertainment | 777.22 | 0.19% |
| Miscellaneous Sales Exp. | 1,811.78 | 0.45% |
| Rent-Carts | 29,810.61 | 7.46% |
| Salaries-Officers | 51,155.00 | 12.80% |
| Salaries & Wages - Employees | 61,156.85 | 15.30% |
| Payrol Tax Exp. | 20,816.36 | 5.21% |
| Rent | 12,400.00 | 3.10% |
| Utilities | 1,692.12 | 0.42% |
| Insurance - General | 40.00 | 0.01% |
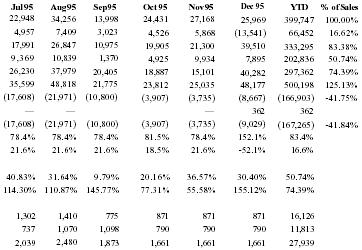
| Jul 95 | Aug 95 | Sep 95 | Oct 95 | Nov 95 | Dec 95 | YTD | % of Sales |
| 22,948 | 34,256 | 13,998 | 24,431 | 27,168 | 25,969 | 399,747 | 100.00% |
| 4,957 | 7,409 | 3,023 | 4,526 | 5,868 | (13,541) | 66,452 | 16.62% |
| 17,991 | 26,847 | 10,975 | 19,905 | 21,300 | 39,510 | 333,295 | 83.38% |
| 9,369 | 10,839 | 1,370 | 4,925 | 9,934 | 7,895 | 202,836 | 50.74% |
| 26,230 | 37,979 | 20,405 | 18,887 | 15,101 | 40,282 | 297,362 | 74.39% |
| 35,599 | 48,818 | 21,775 | 23,812 | 25,035 | 48,177 | 500,198 | 125.13% |
| (17,608) | (21,971) | (10,800) | (3,907) | (3,735) | (8,667) | (166,903) | −41.75% |
| — | — | — | 362 | 362 | |||
| (17,608) | (21,971) | (10,800) | (3,907) | (3,735) | (9,029) | (167,265) | −41.84% |
| 78.4% | 78.4% | 78.4% | 81.5% | 78.4% | 152.1% | 83.4% | |
| 21.6% | 21.6% | 21.6% | 18.5% | 21.6% | −52.1% | 16.6% | |
| 40.83% | 31.64% | 9.79% | 20.16% | 36.57% | 30.40% | 50.74% | |
| 114.30% | 110.87% | 145.77% | 77.31% | 55.58% | 155.12% | 74.39% | |
| 1,302 | 1,410 | 775 | 871 | 871 | 871 | 16,126 | |
| 737 | 1,070 | 1,098 | 790 | 790 | 790 | 11,813 | |
| 2,039 | 2,480 | 1,873 | 1,661 | 1,661 | 1,661 | 27,939 |
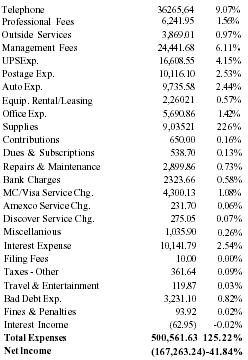
| Telephone | 36,265,64 | 9.07% |
| Professional Fees | 6,241.95 | 1.56% |
| Outside Services | 3,869.01 | 0.97% |
| Management Fees | 24,441.68 | 6.11% |
| UPS Exp. | 16,608.55 | 4.15% |
| Postage Exp. | 10,116.10 | 2.53% |
| Auto Exp. | 9,735.58 | 2.44% |
| Equip. Rental/Leasing | 2,260.21 | 0.57% |
| Office Exp. | 5,690.86 | 1.42% |
| Supplies | 9,035.21 | 2.26% |
| Contributions | 650.00 | 0.16% |
| Dues & Subscriptions | 538.70 | 0.13% |
| Repairs & Maintenance | 2,899.86 | 0.73% |
| Bank Charges | 2,323.66 | 0.58% |
| MC/Visa Service Chg. | 4,300.13 | 1.08% |
| Amexco Service Chg. | 231.70 | 0.06% |
| Discover Service Chg. | 275.05 | 0.07% |
| Miscellanious | 1,035.90 | 0.26% |
| Interest Expense | 10,141.79 | 2.54% |
| Filing Fees | 10.00 | 0.00% |
| Taxes - Other | 361.64 | 0.09% |
| Travel & Entertainment | 119.87 | 0.03% |
| Bad Debt Exp. | 3,231.10 | 0.82% |
| Fines & Penalties | 93.92 | 0.02% |
| Interest Income | (62.95) | −0.02% |
| Total Expenses | 500,561.63 | 125.22% |
| Net Income | (167,263.24) | −41.84% |
Key Ratio Analysis
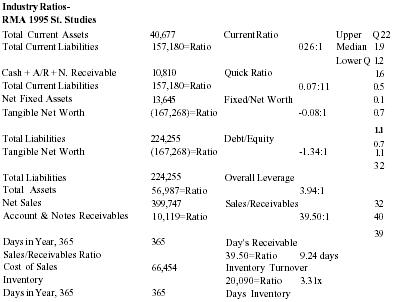
| Statement Dated: 12/31/95 | |||||
| Industry Ratios-RMA 1995 St. Studies | |||||
| Total Current Assets | 40,677 | Current Ratio | Upper | Q2.2 | |
| Total Current Liabilities | 157,180=Ratio | 026:1 | Median | 1.9 | |
| Lower Q | 1.2 | ||||
| Cash + A/R + N. Receivable | 10,810 | Quick Ratio | 1.6 | ||
| Total Current Liabilities | 157,180=Ratio | 0.07:11 | 0.5 | ||
| Net Fixed Assets | 13,645 | Fixed/Net Worth | 0.1 | ||
| Tangible Net Worth | (167,268)=Ratio | −0.08:1 | 0.7 | ||
| 1.1 | |||||
| Total Liabilities | 224,255 | Debt/Equity | 0.7 | ||
| Tangible Net Worth | (167,268)=Ratio | −1.34:1 | 1.1 | ||
| 3.2 | |||||
| Total Liabilities | 224,255 | Overall Leverage | |||
| Total Assets | 56,987=Ratio | 3.94:1 | |||
| Net Sales | 399,747 | Sales/Receivables | 32 | ||
| Account & Notes Receivables | 10,119=Ratio | 39.50:1 | 40 | ||
| 39 | |||||
| Days in Year, 365 | 365 | Day's Receivable | |||
| Sales/Receivables Ratio | 39.50=Ratio | 9.24 days | |||
| Cost of Sales | 66,454 | Inventory Turnover | |||
| Inventory | 20,090=Ratio | 3.31x | |||
| Days in Year, 365 | 365 | Days Inventory | |||

| Cost of Goods/Inventory Ratio | 3.31 = Ratio | 110.34 days | |||
| Net Sales | 399,747 | Sales to Working Capital | |||
| Curr. Assets - Curr. Liabilities | (116,503) = Ratio | −3.43x | RMA Ratios | NA | |
| Net Profit + Depr. + Amort. | (167,268) | Cash Flow/Long Term Debt | |||
| Current Portion L T Debt | 7,000 = Ratio | −23.90x | |||
| Profit, before Tax | (167,268) | Return on Equity | |||
| Tangible Net Worth (Equity) | (167,268) = Ratio | 1.00% | |||
| Profit, before Tax | (167,268) | Return on Assets | |||
| Total Assets | 59,987 = Ratio -2.79 | ||||
THIS PORTION OF PAGE INTENTIONALLY LEFT BLANK SEE NEXT PAGE FOR PROJECTED CASH FLOW TABLE
Projected Cash Flow
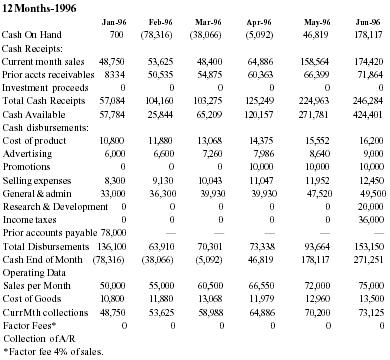
| 12 Months-1996 | ||||||
| *Factor fee 4% of sales. | ||||||
| Jan-96 | Feb-96 | Mar-96 | Apr-96 | May-96 | Jun-96 | |
| Cash On Hand | 700 | (78,316) | (38,066) | (5,092) | 46,819 | 178,117 |
| Cash Receipts: | ||||||
| Current month sales | 48,750 | 53,625 | 48,400 | 64,886 | 158,564 | 174,420 |
| Prior accts receivables | 8,334 | 50,535 | 54,875 | 60,363 | 66,399 | 71,864 |
| Investment proceeds | 0 | 0 | 0 | 0 | 0 | 0 |
| Total Cash Receipts | 57,084 | 104,160 | 103,275 | 125,249 | 224,963 | 246,284 |
| Cash Available | 57,784 | 25,844 | 65,209 | 120,157 | 271,781 | 424,401 |
| Cash disbursements: | ||||||
| Cost of product | 10,800 | 11,880 | 13,068 | 14,375 | 15,552 | 16,200 |
| Advertising | 6,000 | 6,600 | 7,260 | 7,986 | 8,640 | 9,000 |
| Promotions | 0 | 0 | 0 | 10,000 | 10,000 | 10,000 |
| Selling expenses | 8,300 | 9,130 | 10,043 | 11,047 | 11,952 | 12,450 |
| General & admin | 33,000 | 36,300 | 39,930 | 39,930 | 47,520 | 49,500 |
| Research & Development | 0 | 0 | 0 | 0 | 0 | 20,000 |
| Income taxes | 0 | 0 | 0 | 0 | 0 | 36,000 |
| Prior accounts payable | 78,000 | — | — | — | — | — |
| Total Disbursements | 136,100 | 63,910 | 70,301 | 73,338 | 93,664 | 153,150 |
| Cash End of Month | (78,316) | (38,066) | (5,092) | 46,819 | 178,117 | 271,251 |
| Operating Data | ||||||
| Sales per Month | 50,000 | 55,000 | 60,500 | 66,550 | 72,000 | 75,000 |
| Cost of Goods | 10,800 | 11,880 | 13,068 | 11,979 | 12,960 | 13,500 |
| Curr Mth collections | 48,750 | 53,625 | 58,988 | 64,886 | 70,200 | 73,125 |
| Factor Fees* | 0 | 0 | 0 | 0 | 0 | 0 |
| Collection of A/R | ||||||
Projected Annual Financial Statements
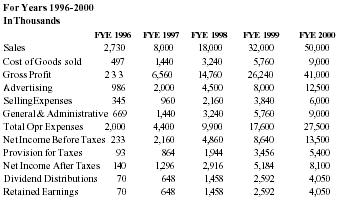
|
For Years 1996-2000
In Thousands |
|||||
| FYE 1996 | FYE 1997 | FYE 1998 | FYE 1999 | FYE 2000 | |
| Sales | 2,730 | 8,000 | 18,000 | 32,000 | 50,000 |
| Cost of Goods sold | 497 | 1,440 | 3,240 | 5,760 | 9,000 |
| Gross Profit | 2,233 | 6,560 | 14,760 | 26,240 | 41,000 |
| Advertising | 986 | 2,000 | 4,500 | 8,000 | 12,500 |
| Selling Expenses | 345 | 960 | 2,160 | 3,840 | 6,000 |
| General & Administrative | 669 | 1,440 | 3,240 | 5,760 | 9,000 |
| Total Opr Expenses | 2,000 | 4,400 | 9,900 | 17,600 | 27,500 |
| Net Income Before Taxes | 233 | 2,160 | 4,860 | 8,640 | 13,500 |
| Provision for Taxes | 93 | 864 | 1,944 | 3,456 | 5,400 |
| Net Income After Taxes | 140 | 1,296 | 2,916 | 5,184 | 8,100 |
| Dividend Distributions | 70 | 648 | 1,458 | 2,592 | 4,050 |
| Retained Earnings | 70 | 648 | 1,458 | 2,592 | 4,050 |
Assumptions

| % Cost of Goods Sold | 18% |
| % Selling Expenses | 12% |
| % General & Administrative | 18% |
| % Tax Provision | 40% |
| Dividends - of NATP | 50% |
| Advertising -1996 | 40% |
| Advertising - all other years | 25% |
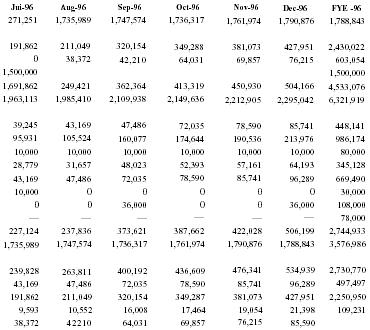
| Jul-96 | Aug-96 | Sep-96 | Oct-96 | Nov-96 | Dec-96 | FYE -96 |
| 271,251 | 1,735,989 | 1,747,574 | 1,736,317 | 1,761,974 | 1,790,876 | 1,788,843 |
| 191,862 | 211,049 | 320,154 | 349,288 | 381,073 | 427,951 | 2,430,022 |
| 0 | 38,372 | 42,210 | 64,031 | 69,857 | 76,215 | 603,054 |
| 1,500,000 | 1,500,000 | |||||
| 1,691,862 | 249,421 | 362,364 | 413,319 | 450,930 | 504,166 | 4,533,076 |
| 1,963,113 | 1,985,410 | 2,109,938 | 2,149,636 | 2,212,905 | 2,295,042 | 6,321,919 |
| 39,245 | 43,169 | 47,486 | 72,035 | 78,590 | 85,741 | 448,141 |
| 95,931 | 105,524 | 160,077 | 174,644 | 190,536 | 213,976 | 986,174 |
| 10,000 | 10,000 | 10,000 | 10,000 | 10,000 | 10,000 | 80,000 |
| 28,779 | 31,657 | 48,023 | 52,393 | 57,161 | 64,193 | 345,128 |
| 43,169 | 47,486 | 72,035 | 78,590 | 85,741 | 96,289 | 669,490 |
| 10,000 | 0 | 0 | 0 | 0 | 0 | 30,000 |
| 0 | 0 | 36,000 | 0 | 0 | 36,000 | 108,000 |
| — | — | — | — | — | — | 78,000 |
| 227,124 | 237,836 | 373,621 | 387,662 | 422,028 | 506,199 | 2,744,933 |
| 1,735,989 | 1,747,574 | 1,736,317 | 1,761,974 | 1,790,876 | 1,788,843 | 3,576,986 |
| 239,828 | 263,811 | 400,192 | 436,609 | 476,341 | 534,939 | 2,730,770 |
| 43,169 | 47,486 | 72,035 | 78,590 | 85,741 | 96,289 | 497,497 |
| 191,862 | 211,049 | 320,154 | 349,287 | 381,073 | 427,951 | 2,250,950 |
| 9,593 | 10,552 | 16,008 | 17,464 | 19,054 | 21,398 | 109,231 |
| 38,372 | 42,210 | 64,031 | 69,857 | 76,215 | 85,590 |
Comment about this article, ask questions, or add new information about this topic: