Chemical Manufacturer
BUSINESS PLAN CHEMALYZE INC.
7201 Rocky Mountain Way
Denver, Colorado 80202
Chemalyze is developing a chemical analyzer and Sensor Cartridge, based upon the Micro-Tongue™ technology, which can instantly analyze complex chemical solutions. Chemalyze's analyzer and Sensor Cartridge will enable economical, real-time analysis of many complex chemical solutions, instantly identifying the presence and quantity of multiple chemicals within a mixture. This plan was provided by Kent Bradshaw, Richard Burgess, Dr. Paul Kunko, and Jason D. Levin; and was compiled in conjunction with the MOOT CORP® competition sponsored by the University of Texas at Austin.
- EXECUTIVE SUMMARY
- CHEMALYZE PROVIDES THE SOLUTION
- MARKETING STRATEGY
- MANUFACTURING STRATEGY
- MANAGEMENT
- FINANCIAL OVERVIEW
- APPENDICES
EXECUTIVE SUMMARY
The Vision
Chemalyze is developing a chemical analyzer and Sensor Cartridge, based upon the Micro-Tongue™ technology, which can instantly analyze complex chemical solutions. This major breakthrough in analyzer technology enables fine chemical manufacturers to:
- Significantly improve product quality
- Increase manufacturing efficiency
- Rapidly respond to processing problems
These benefits will save millions of dollars annually by reducing downtime and improving plant utilization.
In addition, numerous medical applications need real-time, on-the-spot, easy-to-use chemical analysis, which can save lives and reduce expensive hospital laboratory costs.
The Gap in the Market
Many liquid processing industries do not have the technology available for real-time measurement of product quality. The current approach is to remove a sample from the manufacturing process and perform a laboratory test. The time delay in this approach could result in high volumes of product currently in production being rejected, thrown away, or re-processed. These inefficiencies can have a significant impact on a company's bottom line. Product costs can account for 40 percent of sales revenues. The online analyzers currently available are very expensive, have limited applications and accuracy, require skilled operators, and generate delays in providing results.
Meeting the Market's Needs
Chemalyze's analyzer and Sensor Cartridge will enable economical, real-time analysis of many complex chemical solutions, instantly identifying the presence and quantity of multiple chemicals within a mixture. The results can be fed directly into a process control computer, allowing customers to consistently produce high-quality products on time, every time.
Implementation
Marketing and Distribution Strategy
The initial market niche for Chemalyze's analyzer within the U.S. is estimated to be worth greater than $500 million with annual sales of its Sensor Cartridge worth close to $130 million (see Primary Market). Chemalyze intends to enter this market by partnering with large process control original equipment manufacturers (OEMs) that supply instrumentation to fine chemical manufacturers, pharmaceutical companies, and biotechnological companies. Chemalyze's goal is to design, develop, and beta-test analyzers through industrial collaborations and then sublicense the analyzer design to its OEM partner who will manufacture, market, and distribute the analyzers. Chemalyze will ramp up its Sensor Cartridge manufacturing and act as a sole supplier of Sensor Cartridges to the customers of its OEM partners. Other potential markets include but are not limited to food and beverage processing, medical diagnostics, and environmental testing.
Manufacturing Strategy
The analyzer consists of a customized housing into which the Sensor Cartridge can be placed and exposed to liquid chemicals within a process plant. The Sensor Cartridge contains a small silicon chip that holds chemically adapted beads housed in a disposable plastic cartridge. These beads react by producing a color pattern when exposed to different chemicals. Chemalyze will design and produce the chemical receptors to be placed on the beads and purchase the remaining components for the Sensor Cartridge from subcontractors. Chemalyze will NOT assemble the final analyzer instrument but will sublicense to a larger OEM partner. Chemalyze will neither incur the capital costs of developing a manufacturing facility for the instrumentation nor the expense of marketing or distributing the analyzer. However, the design, development, and manufacture of the bead chemistry that makes up the added value provided by the Sensor Cartridge will remain proprietary to Chemalyze.
Financial Strategy
Chemalyze is seeking seed capital of $1,500,000 for initial company operations to:
- Secure the license for the Micro-Tongue™ from the University of Colorado
- Hire a seasoned CEO and management team
- Develop a pre-manufacturing prototype from its proven analyzer technology
- Continue negotiations with potential industrial partners and customers
- Begin alpha-testing with select corporate collaborators
- Fund continuous development of the Micro-Tongue™ within the inventors' labs
Formal operations will begin in the second half of 1999. About one year later, Chemalyze will raise an additional $8 million in private equity, corporate partnerships, and federal grants to:
- Begin full-scale product development (designing novel chemical receptors)
- Commence receptor bead production
- Identify outside vendors and contract the design and manufacture of the Sensor Cartridge
- Develop corporate partnership efforts
Chemalyze intends to raise another $12 million in follow-on investment in the third year of operations to scale-up its chemistry development and new product development efforts. Chemalyze projects $57 million in revenue with $11 million in net income by year six. The initial investor will receive an equity stake in the company and preferential rights to invest in subsequent equity rounds.
Management
Chemalyze was founded by four graduate students from the University of Colorado Graduate School of Business. Its current president and chief executive officer, Roger Dalton, has over 6 years' experience in both academic research and industrial drug development. Roger spent 4 years as a scientist in a start-up gene therapy company. More recently, he worked in business development at a functional genomics company alongside the DNA microchip product development team. He is currently completing his M.B.A. at the University of Colorado.
Chemalyze's vice president and chief financial officer, Joshua Small, is a registered CPA with 5 years of public accounting experience. Joshua spent 3 years as a senior consultant at Citibank, then joined KPMG Peat Marwick as a supervising senior before returning to Colorado to pursue his M.B.A. at the University of Colorado. While completing his degree, he has been working for Trinity Bank, a venture banking firm.
Two of Chemalyze's founders are fulfilling advisory roles. Sidney Pittman, a chemical engineer with a decade of experience in operations and marketing high-tech products, is consulting with Chemalyze as a strategy consultant. Dr. John Barnes has spent the last 8 years performing pharmacology research and is consulting with Chemalyze as a market analyst.
Chemalyze is further strengthened by its collaboration with the four researchers that invented the Micro-Tongue™ at the University of Colorado. These partners are Drs. Brian Boleyn, an associate professor of chemistry and biochemistry, Ernest MacDonald, an associate professor of chemistry, Ian Kirkland, a professor of electrical engineering, and Lee Shu, an assistant professor of chemistry and biochemistry. Each inventor is an active member of Chemalyze's Scientific Advisory Board.
Chemalyze's advisers consist of an impressive group of seasoned businesspeople, including Ronald Mullen, former president of Presario Computers and chairman of the board of LakeTel, Dr. Andrew Rolla, former CEO of Avington Technologies Corporation and Smith Opthalmics; and Jennifer Zweig, president of the Chem.com Group.
The Problem
Many of the products we use every day—from sodas to the fertilizer we put on our lawns to prescription drugs—are made from complex mixtures of many chemicals.
These products are manufactured in sophisticated processing plants. Despite the sophistication of these plants, companies often do not know if the composition of their product is correct until a final laboratory test is performed at the end of the process. If a mistake has been made, the result could be millions of dollars worth of poor quality product that either needs expensive reprocessing or must simply be thrown away (see Primary Market).
Processing companies cannot completely measure product streams during production because the technology currently available for online process analysis consists of basic analyzers that usually measure only one aspect of the product. This information alone is insufficient for highly efficient process control. To truly know the composition of an intermediate or final product, a sample must be removed from the process stream and taken to a laboratory for analysis. These tests take a lot of time, during which high volumes of expensive product could be lost. Hence, there is a tremendous need for chemical manufacturing companies to be able to measure the complex mixtures within their processing plants in a real-time manner. Such technology would enable them to produce high quality products on time, every time.
The Solution
The solution to this gap in the market is at hand. Chemalyze Inc. is partnered with world-class researchers at the University of Colorado who are developing the first Micro-Tongue™. The Micro-Tongue™ is an analyzer that mimics the human tongue using novel chemical microsensor technology. This recent scientific breakthrough in real-time measurement of complex chemical solutions is a vast improvement over current analyzer technology.
The Micro-Tongue™ consists of a light source that shines onto chemically adapted polymer beads arranged on a small silicon wafer, which is known as a Sensor Chip. These beads change color based on the presence and quantity of specific chemicals. Currently the receptors are sensitive to 1 part per million, however, 1 part per billion sensitivity is expected. The color pattern is captured by a digital camera and the resulting video signal is converted into data using a video capture board and a personal computer.
This data output is in a format compatible for input into a process control computer. The polymer beads can be chemically adapted to detect specific chemical compounds within virtually any chemical solution. The capabilities of the technology range from the measurement of simple chemical compounds like calcium carbonate in water (which effects water hardness), to complex organic compounds, such as hemoglobin in blood and proteins in food.
CHEMALYZE PROVIDES THE SOLUTION
The Company
Chemalyze Inc. is a privately-held Delaware "C" corporation operating in the state of Colorado since October 1998, founded by Joshua Small, Sidney Pittman, John Barnes, and Roger Dalton to commercialize the Micro-Tongue™. Chemalyze aims to be the leading niche developer of real-time chemical analyzers to industrial and medical markets within seven years. Chemalyze will provide added value to these markets with real-time, sensitive, and accurate analytical hardware that is on the leading edge of chemical analyzer technology.
The Product Benefits
Chemalyze's initial product is a real-time liquid analyzer focused on the needs of fine chemical, pharmaceutical, and biotechnological manufacturing companies. These companies produce many complex chemical mixtures of high added value in their manufacturing processes. Chemalyze's analyzer can assess the presence and quantity of multiple chemicals in a process mixture, enabling such companies to:
- Measure multiple chemicals simultaneously
- Generate real-time results with laboratory level accuracy
- Use a compact instrument installed onsite
- Convert these results into a digital output suitable for direct input into a process control computer
The key end-user benefits are:
- Consistent quality with less waste
- Improved efficiency and utilization by reducing the quality control bottlenecks
- Increased customer satisfaction from better products without delivery delays
Industry Need for Chemalyze's Technology
Recently, the National Institute of Standards and Technology (NIST) reviewed the field of chemical measurement and identified a multitude of needs and challenges for future development (see this report on NIST's website at http://www.nist.gov/cstl/hndocs/ChemMeasmain.html ). The NIST report recognized the "enabling" capability of chemical analysis for several applications. NIST developed a list of seven key application areas in need of chemical measurement improvements, which included process control and product development, but found the current state of technology to be less than optimal, stating that "real-time analytical measurements are not generally available, either on-line or off-line."
According to the report, the reason most real-world chemical measurements are conducted off-line is that many sophisticated methods of chemical analysis are not finding their way from the laboratory to research and development (R&D) and manufacturing facilities. This system needs to change, based on chemical manufacturers' desires for reduced costs, increased efficiency, increased speed to market and improved product quality. NIST expects the changes to be realized as "robust techniques for real-time, highly reliable analyses in practical environments." These new solutions will be sensors that respond quickly to change in complex manufacturing environments. Current methods, such as Gas Chromatography and Mass Spectrometry, are too slow to adapt to the above dynamic manufacturing trends (see Competition).
The NIST report further defined industry needs against a likely time frame. Short-term needs (less than 3 years) include a systems approach (integrated sampling, online detection, signal transmission, data handling, and maintenance), real-time compositional information for liquid phase streams, increased speed of analysis, sensitivity, and selectivity. Medium and long-term needs include high reliability, a PC-based generic user interface, real-time trace analysis, and miniaturization. All of these needs are addressed by Chemalyze's Micro-Tongue™ technology.
Market Analysis
Sales of process control systems are experiencing strong growth. Sales in 1997 were $3.86 billion and are expected to reach $4.6 billion by the year 2000 and $5.75 billion by 2004. The chemical analyzer industry is slated for rapid expansion, growing at an annual rate of 7.8 percent from $1.45 billion in 1991 to $3.10 billion in 2001. This market is split almost evenly between liquid and gas analysis. Nearly half this total market is for process control. Within this market, revenues for online analyzers will grow at a similar rate and should reach $1.3 billion in 2000, up from $880 million in 1993 ( Chemical & Engineering News , March 1995, Vol. 73, 11, p. 25). Typical industries that use process control analyzers are:
Process Industries
- Fine chemicals
- Chemical intermediaries
- Pharmaceuticals
- Biologicals
- Medicinals
- Detergents
- Fertilizers
- Soft drinks, Spirits, Malt beverages
- Explosives testing
- Paints and dyestuffs
- Food flavorings
- Personal care products (e.g. fragrances and cosmetics)
Clinical Diagnostic Systems
- Diagnostic testing
- Point-of-care testing
Environmental Management Systems
- Monitoring water quality
- Waste stream management
Chemalyze's primary market is process control analyzers for the fine chemicals industry. After Chemalyze has developed a strong presence in this market it will broaden its R&D efforts and marketing efforts to include medical diagnostics. Chemalyze will assess other market opportunities on a case-by-case basis and focus on those industries that are the most price-insensitive and where Chemalyze can quickly deliver the most value and capture the most profit.
Primary Market
Worldwide prescription pharmaceutical sales reached $265 billion in 1997, and the market is continuing to grow. However, profit margins are threatened by strict industry requirements on processes, ever changing government regulations, and increasing competitive pressures. In response, pharmaceutical companies are spending increasing amounts on R&D to combat rising manufacturing costs, which are currently estimated at 24 percent of sales ("The Drug Industry and PBM companies in 1995," Cool, K.). Of the $21.1 billion spent on R&D in 1998, 9 percent was spent on process improvements totaling $1.91 billion ( Pharmaceutical Researchers & Manufacturers of America , 1998 Industry Report). The process improvement investments are classically price-insensitive. Pharmaceutical manufacturers have indicated that Chemalyze's technology could provide an opportunity to improve their processes by significantly reducing manufacturing costs. This can be achieved by improving upon current quality control methods and relieving chemical analysis bottlenecks within the process.
The market opportunity for Chemalyze's technology among pharmaceutical manufacturers has been determined by assessing the process control requirements of the small, medium, and large manufacturers in the U.S. today. Of the 700 pharmaceutical firms in the U.S., we have estimated that 35 percent of those companies have small process plant manufacturing facilities, 35 percent have medium facilities, and 30 percent have large manufacturing plants. To identify the pharmaceutical manufacturer's return on investment from the introduction of Chemalyze's technology we estimated its impact of these different size plant.
Small Process Facility
Based upon discussions with senior managers at biopharmaceutical manufacturers, Chemalyze estimates that 15-20 percent of a small facility's capacity is lost yearly from product quality problems at a cost of $250,000. With Analyzer 1 in place on one plant line, a firm of this size would save approximately $180,000/year by reducing total number of lost batches by 66 percent. With Analyzer 2 in place, such a firm would save approximately $60,000/year by eliminating one lab technician needed to perform Quality Control (QC) experimentation for Food and Drug Administration (FDA) compliance. Thus, the total savings estimated per small plant line are approximately $240,000 per year.
If a small process manufacturing facility has 2 plant lines that need 2 analyzers each, as well as having one pilot plant facility which may use 1 analyzer, then the total number of analyzers for a small process facility is equal to 5 analyzers, with a total savings of $480,000.
Medium Process Facility
Chemalyze has estimated that a medium facility will have a similar production scheme as a small facility only it would include 6 plant lines rather than 2. Assuming each of 6 lines requires 2 analyzers—adding 3 extra analyzers for a larger pilot plant—the total number of analyzers for a medium process facility is equal to 15 analyzers, with a total savings of $1,440,000.
Large Process Facility
A large process facility would have a more complex system for production and would work on a much larger scale than both a small and medium facility. Chemalyze has estimated that a large process facility with 1 plant line utilizing 4 feed lines to produce one final product could utilize 5 Chemalyze QC analyzers. This total does not include additional analyzers necessary for pilot scale experimentation. If we assume that a large facility has a throughput volume 10 times that of a small facility and Chemalyze's analyzers can similarly prevent 66 percent of the 15-20 percent lost batches, then the savings, if linearly scaled, would be approximately $2 million per annum.
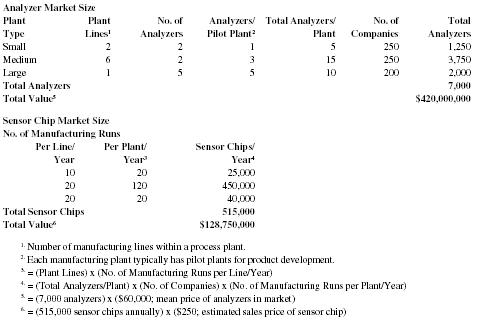
Market Size
The estimated size of the pharmaceutical manufacturing market niche for Chemalyze's Micro-Tongue™ technology (see also Marketing Strategy for details on industry structure) is shown in the following table.
| Analyzer Market Size | ||||||
| Plant Type | Plant Lines 1 | No. of Analyzers | Analyzers/Pilot Plant 2 | Total Analyzers/Plant | No. of Companies | Total Analyzers |
| 1. Number of manufacturing lines within a process plant. | ||||||
| 2. Each manufacturing plant typically has pilot plants for product development. | ||||||
| 3. = (Plant Lines) x (No. of Manufacturing Runs per Line/Year) | ||||||
| 4. = (Total Analyzers/Plant) x (No. of Companies) x (No. of Manufacturing Runs per Plant/Year) | ||||||
| 5. = (7,000 analyzers) x ($60,000; mean price of analyzers in market) | ||||||
| 6. = (515,000 sensor chips annually) x ($250; estimated sales price of sensor chip) | ||||||
| Small | 2 | 2 | 1 | 5 | 250 | 1,250 |
| Medium | 6 | 2 | 3 | 15 | 250 | 3,750 |
| Large | 1 | 5 | 5 | 10 | 200 | 2,000 |
| Total Analyzers | 7,000 | |||||
| Total Value 5 | $420,000,000 | |||||
| Sensor Chip Market Size | ||||||
| No. of Manufacturing Runs | ||||||
| Per Line/Year | Per Plant/Year 3 | Sensor Chips/Year 4 | ||||
| 10 | 20 | 25,000 | ||||
| 20 | 120 | 450,000 | ||||
| 20 | 20 | 40,000 | ||||
| Total Sensor Chips | 515,000 | |||||
| Total Value 6 | $128,750,000 | |||||
Based upon the previous set of assumptions for the number of QC analyzers that a given pharmaceutical manufacturing firm would use our calculations indicate a national market for approximately 7,000 analyzers. However, analyzer system sales represent only one source of revenue. Chemalyze's technology incorporates a disposable element (a Sensor Cartridge), that needs to be replaced after each manufacturing run. As a result, the maximum Sensor Cartridge sales for this market would total over .5 million units per year. These calculations assume a relatively low number of analyzers per plant and ignore the many contract pharmaceutical manufacturers, who will also have multiple applications for Chemalyze's technology.
It is also important to note that this market estimation only considers the number of available plant lines in the pharmaceutical industry within the U.S. and does not take into account the number of plant lines in biotechnology and other types of fine chemical processing facilities(e.g. producers of chemical intermediaries to supply other manufacturers and users). In fact, large pharmaceutical companies spend 16 percent of their R&D budget (approximately $3 billion) on outsourcing pharmaceutical development and manufacturing to contract pharmaceutical manufacturers ("The Pharma Giants: Ready for the 21st Century?", Eagan, R., Hayes, R., 1998). Chemalyze expects that when the other potential buyers in the fine chemicals market are evaluated that the total market within the U.S. for Chemalyze's analyzers and Sensor Cartridges will increase substantially.
Secondary Market
The most attractive future market for Chemalyze's technology, once the technology has been fully developed, is the Point-of-Care (POC) medical diagnostics market. Medical diagnostics consists of the detection of substances in body fluids (e.g. real-time HIV testing). Fast and accurate analyses are essential for emergency medicine and high quality patient care. The estimated worldwide market for medical diagnostics in 1997 was $19.5 billion. These revenues are generated primarily from testing in hospitals and commercial labs, which account for $15.5 billion of the medical diagnostics market (POC Testing/IVD - Industry Report, Sutro & Co. Inc., 1/29/98).
Current federal regulations specify extensive and expensive training for commercial laboratory personnel. These labs are a significant source of hospital overhead. A handheld version of Chemalyze's technology will provide nurses and physicians with on-the-spot analyses, saving lives and eliminating much of the expense of laboratory testing.
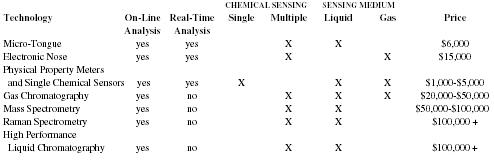
Competition
Analyzer technologies that compete with Chemalyze's Micro-Tongue™ are:
- Physical property meters and single chemical sensors
- Gas chromatography (GC)
- Mass spectrometry (MS) and Raman spectrometry
- High performance liquid chromatography (HPLC)
These technologies either have severe limitations in the complexity of the chemical solutions they can analyze or they do not provide real-time analysis. Given the Micro-Tongue's™ capabilities these technologies are considered less of a direct threat and more complementary to Chemalyze. This is because its analyzer can be associated with these existing systems to provide another dimension on their current analytical capabilities.
Competing Technologies
1. Physical property meters and single chemical sensors
A sensor system designed to measure pH, conductivity, or to identify a single chemical in a solution can only measure one aspect of the process. A pH meter only tells you if your solution is acidic or alkaline. An ion conductivity probe only tells you of the electrical conductivity of your solution. Gas sensors only tell you how much of a particular gas, e.g. dissolved oxygen, is in your solution. Even the systems that companies claim can sense multiple chemicals have severe limitations, both in the number of chemicals detected and type of chemical that can be detected. This contrasts with Chemalyze's technology, which enables the user to sense multiple chemicals in a complex solution simultaneously, and in real time.
2. Gas Chromatography (GC)
GC vaporizes a liquid sample and identifies different compounds through their molecular weight. It is mainly used to test for purity. This technology can be used online but requires an additional $20,000 to $50,000 expenditure (on top of a $20,000 initial capital equipment cost) and does not enable real-time data acquisition and analysis, as it takes 5 to 10 minutes to analyze a sample. However, with the assistance of a highly trained operator, GC can analyze virtually any chemical mixture.
3. Mass Spectrometry (MS)
MS uses ionization technology to analyze the liquid sample. It is a laboratory based, expensive (ranging from $50,000 to $150,000) and complicated method for identifying compounds in a mixed solution. However, MS technology has also been in use for a number of years and has established itself as the benchmark in some industries for a limited range of chemicals. It has been used online but requires a few minutes to analyze the samples and eliminates the opportunity to evaluate data in real time. The key disadvantage with MS technology is that it cannot be used with water-based solutions, which are very common in pharmaceutical production.
4. Raman Spectrometry
The latest spectrometry technology is known as Raman spectrometry. It is not fully established as an online process analysis technology. Raman measures the scatter of light by different compounds and can effectively monitor compounds in water based solutions. The main disadvantage is its limited sensitivity, although this may improve over time.
5. High Performance Liquid Chromatography (HPLC)
HPLC uses separation techniques based upon molecular weight to identify chemical compounds. HPLC systems occupy the price range in between GC and MS technology at $100,000 each. HPLC methods are often slow and expensive to scale up and cannot distinguish between similar chemical compounds.
The closest comparable product to Chemalyze's analyzer is an HPLC unit, which is online in pharmaceutical manufacturing environments but cannot provide real-time results. In addition, the results from an HPLC unit need to be interpreted by a highly trained operator. Chemalyze's system provides easily understandable output that can feed directly into a process control computer or a standard PC. However, a new class of companies is developing electronic "nose" technology for analyzers to detect multiple chemical compounds in gaseous form; these companies present a more serious threat. Chemalyze has identified three companies in this space—Aromascan, Inc., Cyrano Sciences, Inc., and Illumina, Inc.
The scientific principles of the electronic nose are similar to the Micro-Tongue™, but are applied to vapors rather than liquids. This particular difference is where Chemalyze's Micro-Tongue™ has an advantage in liquid process control environments or any liquid-based chemical analysis. The polymers that are used for the electronic nose are not effective when immersed in a liquid solution, as they absorb liquid at a significantly slower rate than the polymers used in the Micro-Tongue™. Also, the fragile construction of most electronic nose analyzers makes them impractical for use in liquid process streams.
Competing Multi-Chemical Sensor Companies
- Illumina Inc. is a start-up located in San Diego, California, developing "electronic nose" technology where latex beads are arrayed on the end of a fiber optic bundle. These beads fluoresce in response to the chemical composition of the vapor they are exposed to. A light source sends light down the length of the bundle and the bead's fluorescence is registered by a digital camera. Illumina is targeting the genomics industry's drug discovery and development efforts. Its technology cannot be used in a liquid stream due to the fragile construction of the fiber optic head, and can only be used with vapors under very mild process conditions. They recently raised $750,000 in seed capital and potentially $8 million in follow-on investment from CW Group and ARCH Ventures.
- Cyrano Sciences Inc. is a start-up located in Pasadena, California, developing a handheld electronic nose. When the nose comes in contact with a vapor the polymer receptors react by swelling and electrodes translate this activity into a digital impulse, which is analyzed by computer. The prototype is slightly larger than a handheld calculator. Cyrano expects to sell each instrument for $5,000 and each chip for a few dollars. Currently, Cyrano's technology deals only with vapors. They are focusing on process control systems with intent on applying its technology to medical diagnostics in the future. Cyrano recently raised $3 million in seed capital and obtained $9 million more from J&J Development Corporation, Marquette Venture Partners, and Oak Investment Partners.
- Aromascan, Inc. represents the oldest commercial effort in developing electronic nose technology. It sells a benchtop analyzer that requires individual preparation of each sample to be placed into test tubes in a rack that is then systematically rotated through the sensor system. Aromascan is a publicly held company operating in the United Kingdom. They currently sell four instruments ranging from $30,000 for a benchtop system to $70,000 for an autosampler. The company has been in operation for 4 years and is focusing primarily on the polyurethane process industry and the personal care products industry.
Comparison of Competing Technologies
| CHEMICAL SENSING | SENSING MEDIUM | ||||||
| Technology | On-Line Analysis | Real-Time Analysis | Single | Multiple | Liquid | Gas | Price |
| Micro-Tongue | yes | yes | X | X | $6,000 | ||
| Electronic Nose | yes | yes | X | X | $15,000 | ||
| Physical Property Meters | |||||||
| and Single Chemical Sensors | yes | yes | X | X | X | $1,000-$5,000 | |
| Gas Chromatography | yes | no | X | X | X | $20,000-$50,000 | |
| Mass Spectrometry | yes | no | X | X | $50,000-$100,000 | ||
| Raman Spectrometry | yes | no | X | X | $100,000 + | ||
| High Performance | |||||||
| Liquid Chromatography | yes | no | X | X | $100,000 + | ||
MARKETING STRATEGY
Overview
Chemalyze intends to focus its initial efforts on applying the Micro-Tongue™ technology to the pharmaceutical process control industry and build market share within that specific niche.
Chemalyze will then broaden the scope of its market strategy to include the medical diagnostics market. This expansion will be achieved through the initial design, development, manufacture and sale of handheld, point-of-care blood and urine analyzers.
Primary Customer Analysis & Entry Strategy
Pharmaceutical manufacturers develop new drugs and other pharmaceutical products through laboratory research and development programs. Such product development flows from a laboratory process to mass production via the construction of a small scale manufacturing plant (known as a pilot plant). This pilot plant is used for further experimentation and process optimization, before the company completes the conceptual design of the full-scale plant.
By adopting Chemalyze's analyzer technology at the pilot plant stage, pharmaceutical companies will realize immediate savings through developing a more efficient design. These savings will be multiplied in new and existing full-scale plants from both improved efficiencies and reduced reprocessing costs (see Primary Market). Chemalyze's technology will also improve project economics for conceptual projects. This will make larger plants significantly more cost effective and profitable, thus making some projects viable that were previously uneconomical.
There is an immediate need where Chemalyze's analyzer technology can be applied: every pharmaceutical facility must clean its tanks and pipes. Furthermore, the FDA requires that each facility monitor and document the amount of detergent leftover in the process equipment after each cleanin—referred to as residuals. These residuals have to be tested to assure that they are not contaminating the final product that is packaged and sold on the market. The current cleaning process requires that the system be shut down and the inside of the tanks swabbed. These swabs are then sent to a laboratory that performs an analysis to determine the amount of residual left in the tank or pipe after cleaning. This process takes time and requires that the facility be shut down 1-3 days. Chemalyze can provide a real-time analytical system to monitor the amount of residual left in each plant line, which decreases manufacturing down time and improves plant utilization. Such improvements can be worth far more than the savings detailed in Primary Market when one takes into account the market value of the final product and the lost opportunity cost associated with failed batches. Furthermore, Chemalyze's technology can be utilized at the end of the process as a final QC for lot release testing to test for any trace elements that may be carried over in the product processing. In some of our discussions with process plant operators it became clear that there are not only bottlenecks within the process but that there are also bottlenecks at the final QC release test.
Strategic Alliances
To achieve its vision, Chemalyze will partner with corporations that have distinctive competencies in the development and manufacture of chemical analyzer systems. These companies include:
- Leading liquid process automation and control specialists. These companies can provide1) technical collaboration to enable Chemalyze to develop an analyzer that complements their control systems, 2) capital resources to fund development costs, and 3) well-established distribution channel to pharmaceutical manufacturers. In return, Chemalyze will become an exclusive supplier of Sensor Cartridges to the selected partner, giving the control specialist a significant competitive advantage in process quality control of complex chemical processes.
- Major manufacturers of chemical analyzer (e.g. HPLC) equipment. Chemalyze's analyzer will be a complementary product that expands the capabilities of their existing chemical analyzer systems.
Core Competency
Chemalyze's core competency is in developing real-time analytical platforms for complex chemical solutions. It is not a high volume instrumentation manufacturer, but rather a company that develops customized analytical sensing technology for different industrial applications. Hence, when Chemalyze has developed a pre-production prototype analyzer it will license this product to a major instrument manufacturer and will outsource the design and manufacture of the Sensor Cartridges. However, Chemalyze will continue to develop the Sensor Beads (for residuals testing) and customized beads (for a customer's proprietary chemistries) for its clients. Thus, the core of the proprietary Sensor Cartridge technology (the Sensor Beads) will continue to be developed and manufactured i- house. See Manufacturing Strategy for further detail.
Expansion Strategy into Secondary Markets
Chemalyze will exploit its experience in the fine chemicals industry to leverage its technological competence in other markets that can benefit from multi-chemical sensing. Sensor Cartridges and the Sensor Beads will then be customized for these new markets, again with licensing granted to a large-scale manufacturer when a pre-production prototype analyzer has been developed.
Medical diagnostics is a major secondary market for the company due to its need for real-time chemical analysis of complex chemical solutions. Chemalyze will work toward developing the Micro-Tongue™ into a handheld device that is capable of performing blood and urine analyses in the emergency room. In discussing this opportunity with emergency room physicians and clinical laboratory nurses it has become clear that there is a need for fast response chemical analysis in the emergent care environment to help save lives. Hospital management will also have a keen interest in Chemalyze's technology, as it will result in significant savings in laboratory overhead costs and the resulting reduction in personnel.
Sales Strategy
Pricing: Our initial estimates of the price sensitivity of the primary market for the Micro-Tongue™ show that our potential customer base spends anywhere between $25,000 and $150,000 for sophisticated analyzers. Chemalyze will not only compete on price but will differentiate itself from the current state-of-the-art by offering customized products designed to suit the particular needs of the customer. Chemalyze has estimated a product price of $60,000 per analyzer unit for financial projection purposes. Sensor Cartridges, which will need to be replaced after each manufacturing run, will be priced at $250 per unit.
Positioning: Chemalyze will position the Micro-Tongue™ technology as a leading edge technology for fine chemical process and quality control. It complements existing chemical analysis technology. In the case of HPLC, Chemalyze's analyzer can be adapted to work with existing instrumentation and add to the current analytical methods. Thus, providing a fourth dimension chemical analysis [specific to the chemical(s) of interest and highly selective]. It will also be developed into a cost-effective, highly accurate handheld instrument for performing medical diagnostics in emergency care environments.
Promotion: Chemalyze will sublicense the analyzer to OEM partners and co-development customers. The partners will be responsible for mass marketing the analyzers and pushing the technology through their channels. Chemalyze will identify and focus on a key player in each respective market niche that would benefit from Chemalyze's technology and prosper from the unfair competitive advantage that this selective technology offers.
Chemalyze will work closely with its inventors, supporting the research needed to develop chemical receptors for later markets. As well, it will support the publishing of these efforts in prestigious research journals to position the product as a breakthrough in chemical analyzer technology.
An informational website ( http://www.chemalyze.net ) is in place and a product hotline will be set up to promote awareness and support new and prospective customers. Supporting promotional material such as video demonstrations of the technology and an exhibition demonstration model will also be developed. Acceptance of the technology by fine chemical manufacturing companies will result in the analyzer being adopted for use in pilot plants, quality control laboratories, specified in new process plants and for retrofitting on existing plants. In part, this approach implements a pull strategy, whereby the Process R&D groups for manufacturers (the early adopters) will adopt the technology in the early stages of development and specify the analyzer in their scale-up designs for use in the large scale process facility.
Place: Instrument distribution will be through an OEM that has successfully penetrated the markets that Chemalyze sees as its early adopters. This approach gives Chemalyze's technology access to established and accepted buying channels. The Sensor Cartridges will be shipped direct from Chemalyze to the OEM partner's customers using standard shipping subcontractor services such as UPS, DHL, and Federal Express.
Service and Warranties
Chemalyze will deal with faulty Sensor Cartridges by replacing them directly. The system designed by the OEM partner will be modular, making removal and replacement of hardware parts simple and fast. Thus, whenever a system fails or breaks, the OEM partner can ship a replacement part directly to the customer. This feature allows the unit to be serviced by the client's personnel, thus reducing the need for a large customer service staff. However, Chemalyze's partner may choose to support the analyzer using their own service systems and warranty programs.
MANUFACTURING STRATEGY
Overview
The manufacturing process will primarily consist of in-house chemical receptor R&D and Sensor Bead production. An early stage pre-production prototype analyzer will be developed at Chemalyze but no assembly lines or systems will be created to support instrumentation manufacturing.
When the pre-production prototype is completed, volume manufacturing, marketing, and distribution of the analyzer will be licensed to an expert from whom Chemalyze would expect an 8-10 percent royalty from gross revenues. Chemalyze will design a receptor bead manufacturing facility to produce the beads for the Sensor Cartridges. The polymer beads that are chemically impregnated to make the Sensor Beads can be readily purchased from a number of polymer resin manufacturers for pennies per 1,000 (e.g. Novabiochem sells 450,000 beads for $170). The etched silicon chip can be purchased from a number of micro-electromagnetic systems (MEMS) suppliers (e.g. one such vendor, AMMi, quoted a price of $6 per 1cm2 silicon chip for a 10,000 unit order and a $3 cost per chip for a 250,000 unit order). The Sensor Cartridge can be designed by an outside design firm and the manufacturing can be subcontracted to plants overseas to reduce parts costs.
Initially, Chemalyze intends to develop Sensor Cartridges for many applications across the fine chemicals process industry, such as residuals testing for detergents and other trace elements. Eventually, Chemalyze will provide the R&D effort and customization of the Micro-Tongue™ to fit its clients' particular process needs—conceivably creating partnerships that protect each firm's proprietary chemistry.
Although the polymer beads in their current state are reversible and can be reused, Chemalyze intends to first market the Sensor Cartridge as disposable elements to avoid issues of recalibration. As the technology develops and Chemalyze improves upon its product R&D, the Sensor Cartridges will be designed to be reusable, to provide the client added value, and increase the barrier to entry from competition. This development may also provide an opportunity to increase Sensor Cartridge prices.
Manufacturing Process
The key stages of the manufacturing process are as follows:
- Develop beads for insertion into Sensor Cartridge. Chemalyze's development team will construct the necessary chemical receptors needed for the Sensor Beads to react to the specific chemical compounds identified through initial product marketing to pharmaceutical companies. A batch of beads will then be chemically impregnated. Chemalyze's development team will also work directly with specific corporate partners to identify proprietary chemistries that are specific to the partner and build receptors for their specific use.
- Produce Sensor Chips. The beads will then be inserted into pre-formed wafers (purchased from a MEMS vendor) using a "pick and place" machine in a "clean" environment by a subcontractor.
- Assemble Sensor Cartridge components. The newly produced chips will be encased in a disposable removable cartridge (designed and manufactured by a subcontractor) that can be inserted into the analyzer body (designed by the OEM), which consists of a light source and a CCD chip (to capture the image) enclosed in an insulated casing. A video capture board (to convert the analog visual signal into data) and the relevant software will also be included in the final packaging of the analyzer provided by the OEM partner.
Chemalyze will develop a list of preferred vendors for each outsourced component to assure security of supply. In addition, Chemalyze will carry sufficient safety stock of critical components. Chemalyze believes that the polymer beads, silicon wafers, and plastic cartridge housing will be relatively inexpensive on a large scale and will not add significantly to inventory levels.
Chemalyze's suppliers will not represent a competitive threat to Chemalyze, as each vendor will only be involved in providing a specific aspect of the product, and will be chosen so that they do not have the capability or market exposure to develop a competitive product.
Chemalyze's offices, laboratories, manufacturing, and storage will need to be located in at least a 15,000-square-foot facility. If such a facility doesn't readily exist in Denver, Chemalyze is prepared to work with design engineers, architects, and a builder to build-to-suit as a last resort. Chemalyze is pursuing the option of co-opting with another local start-up that will need lab space and HVAC systems, in order to share overhead costs.
Management
Chemalyze's management team is highly motivated and talented, with complementary skills and experience. However, Chemalyze recognizes that the team needs additional expertise and skills to grow the company rapidly into a market leader. Thus, Chemalyze intends to actively recruit a seasoned CEO, chief scientific officer, and vice president of product marketing to help fill in the gaps of the current management team.
Chemalyze's president and chief executive officer, Roger Dalton, has worked for two start-up biotechnology companies. Most recently, Roger worked for ABC Genes, Inc. in business development, focusing on the market analysis and product development strategy for an emerging DNA microchip technology. Roger will be responsible for licensing the technology from the UT system, as well as coordinating the business development and developing a long-term strategy for corporate growth at Chemalyze.
Chemalyze's vice president and chief financial officer, Joshua Small, will play a key role in securing financing for the company. He brings significant financial and accounting experience to Chemalyze, Inc. having worked as a senior consultant with Citibank. Currently, Joshua is working for Trinity Bank, where he has gained relevant experience working with venture capital firms and venture-backed companies. Joshua is a CPA licensed in Colorado.
In addition, Chemalyze is partnered with the inventors of the Micro-Tongue™ technology at the University of Colorado. Ernest MacDonald, Ph.D. is an associate professor of chemistry and is responsible for combining the organic synthetic chemistry developed by Brian Boleyn, Ph.D., associate professor of chemistry, micro-machining techniques developed by Ian Kirkland, Ph.D., associate professor of electrical engineering, and the detection methodology developed by Lee Shu, Ph.D., assistant professor of chemistry to create the Micro-Tongue™. Chemalyze has been working closely with this multi-disciplinary team of world-class scientists to commercialize the Micro-Tongue™. The inventors will remain actively involved in the creative development of Chemalyze and are eager to support Chemalyze's product development into the future.
Chemalyze's advisers consist of seasoned businesspeople that are bringing their complementary skills to bear on the hurdles that Chemalyze faces. These advisers include Mr. Ronald Mullen, former President of Presario Computers and chairman of LakeTel, Dr. Andrew Rolla, former CEO of Avington Technologies Corporation and Smith Opthalmics, and Ms. Jennifer Zweig, president of the Chem.com Group.
Chemalyze has also been working with a team of consultants to develop its marketing strategy and build upon its understanding of the potential for the Micro-Tongue™ within different commercial markets. Sidney Pittman is a chartered chemical engineer and has 10 years of industry experience, primarily in small technology companies selling to large blue chip corporations such as Mobil Oil and Phillips Petroleum. His experience also includes four years in plant design and operations. Sidney will aid in developing manufacturing and marketing strategies for Chemalyze, Inc. John Barnes, Ph.D., will help identify specific product market opportunities that utilize his scientific strengths developed during his research work in pharmacology, as well as his product development experience with St. Luke's Medical, Inc. Both Sidney and John are co-founders of Chemalyze, Inc. Roger, Joshua, Sidney, and John currently hold 100 percent ownership of the company.
Regulatory Environment
In order to develop and market process control technology to the pharmaceutical industry, Chemalyze will have to provide experimental evidence that shows its technology is robust, reliable, sensitive, and reproducible enough to be used in closely monitored manufacturing environments. However, there are no regulatory compliance issues that Chemalyze would have to address prior to marketing its technology in pharmaceutical process control applications. The customer would be required to validate the use of the instrument and provide the Food and Drug Administration (FDA) with relevant documentation to this effect. Chemalyze will develop protocols with the customer to support the FDA approval process. The FDA's main concern is reproducibility and efficacy. Chemalyze already has data demonstrating the consistency and accuracy (e.g. reproducibility) of its technology.
With regard to marketing Chemalyze's technology for use within hospitals in the U.S., Chemalyze would have to gain regulatory approval through the FDA. The Code of Federal Regulations (CFR) provides guidelines for both the classification of medical devices, as well as the regulatory guidelines necessary to gain approval for the marketing of such devices. Since there is very little way of determining to what extent Chemalyze's instruments will require FDA approval until the instrument is actually designed and built, we will assume that the instrument will require the maximum testing and regulatory approval for a Class II medical device.
Chemalyze will make every effort to remain in contact with the FDA during the period of early development and testing to identify what guidelines Chemalyze needs to follow to obtain regulatory approval. This is a straightforward process and Chemalyze does not consider the regulatory process to be a critical issue.
Intellectual Property
The University of Colorado has filed 2 provisional patent applications with the United States Patent and Trademark Office (USPTO). The first office action for the first provisional application was recently submitted to the USPTO by the university's lawyers. The university, its lawyers, and Chemalyze believe that a patent for this technology could be issued as early as June of 2000. We are working with the Office of Technology Licensing and Intellectual Property for the University of Colorado system to obtain a favorable license to this technology for our specified fields of use. We have the full support of the University of Colorado researchers who invented this technology.
FINANCIAL OVERVIEW
Seed Stage: Present - Jan. 2000
Chemalyze is currently in the concept stage and has several milestones to reach before it begins full operations. We plan on achieving the following goals by the end of 1999:
- Obtain the license for the technology from the University of Colorado
- Perform research on detailed product specifications
- Build a pre-manufacture prototype for one application
- Establish a joint venture or co-development arrangement with at least one corporate partner
- Begin making Sponsored Research Agreement (SRA) payments to University of Colorado to maintain R&D efforts
- Hire a seasoned CEO
- Hire veteran product development scientists
- Hire a vice president of product marketing
In order to achieve these milestones, the company is seeking $1,500,000 investment from an investor or group of investors in exchange for an equity share of the company. The seed investors will have preferential rights in subsequent equity rounds.
Start-up Stage: Feb. 2000 - Feb. 2001
Once the above milestones are met, Chemalyze will require $8,000,000 in start-up capital. The company plans on obtaining these funds from venture investors and/or a joint venture with a corporate partner. It is also pursuing SBIR (Small Business Innovation and Research grants) funding and NIST ATP (National Institute of Standards and Technology Advanced Technology Program grant) awards to supplement the equity capital raised in the prior round.
The funds will be used to purchase the capital equipment needed to start operations and to hire relevant personnel to develop the beta versions of the product. (See Financial Statements for a list of staffing and capital expenditures, respectively).
The milestones for completing the start-up stage are:
- Hire the senior management, relevant executives, and other staff. This will include more industrial scientists to continue Sensor Cartridge development, and the requisite support staff.
- Build out the first phase of a facility.
- Purchase equipment to complete the R&D laboratory and set-up receptor chemistry manufacturing.
- Identify vendors to design and manufacture the Sensor Cartridge (includes arraying beads).
- Identify and begin beta testing with companies in the pharmaceutical industry. This step will be achieved with the support of our corporate partner and is the beginning of our revenue stream.
- Identify and create a corporate partnership agreement with an OEM to manufacture, market, and distribute Chemalyze's analyzers.
Development Stage and Product Launch: Feb. 2002 and onward
At this point we will have completed successful beta testing of our product. We will ramp up our manufacturing operations of the Sensor Cartridge. Our OEM partners will start a full-scale marketing campaign. This stage will require another round of financing for additional personnel, laboratory and manufacturing equipment, and for working capital purposes. A final $12,000,000 round of financing is needed before Chemalyze generates positive cash flows in 2004.
Exit Strategy
Chemalyze will have healthy levels of revenue and net income in FY 2004 and FY 2005. At this point, investors should be able to achieve liquidity through an initial public offering or through acquisition by a company in the process control industry.
Critical Risks
Licensing and cross-licensing
Chemalyze must license the Micro-Tongue™ technology from the University of Colorado. Chemalyze has the backing of the inventors of the technology and has a high probability of gaining a license for the technology within specified fields of use. Chemalyze will organize itself and implement strategies to protect itself and proactively avoid operating within the scope of issued patents thus, preventing infringement lawsuits from the current competition.
A patent for the Micro-Tongue™ must be issued for Chemalyze to operate.
Chemalyze will retain the services of an intellectual property (IP) law firm to perform an infringement study on patent USPTO #4,312,700 (technology developed at Tufts University and licensed exclusively to Illumina, Inc.) and USPTO #4,350,405 (technology developed at Caltech and licensed exclusively to Cyrano Sciences, Inc.). Both patents are issued for electronic nose technology. However, Chemalyze feels that honest diligence is necessary to confirm its freedom to operate.
If, after a patent is issued for the Micro-Tongue™ and Chemalyze obtains a license from the University of Colorado, its attorneys feel that there are issues around possible infringement (either against Chemalyze's property or by Chemalyze), Chemalyze will develop cross-licensing relationships with Illumina and Cyrano. This action serves the interest of all parties by enabling each the freedom to operate and eliminating the likelihood of costly legal entanglements in the future.
To further differentiate its IP portfolio and improve its ability to protect its technology, Chemalyze will continue to expand its IP portfolio by patenting its novel chemical receptors and Sensor Cartridge design.
This page left intentionally blank to accommodate tabular matter following.
APPENDICES
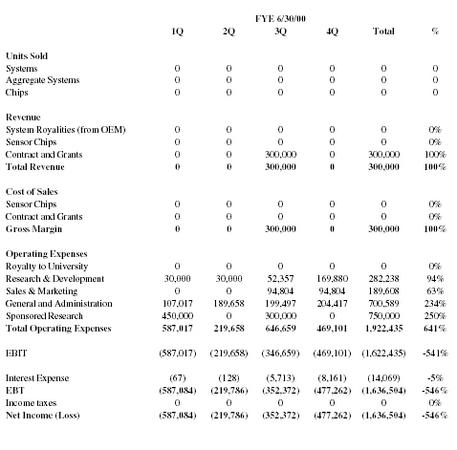
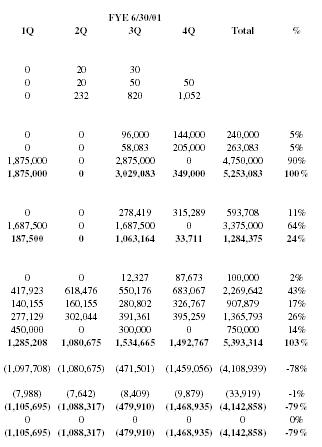
Income Statement
| FYE 6/30/00 | ||||||
| 1Q | 2Q | 3Q | 4Q | Total | % | |
| Units Sold | ||||||
| Systems | 0 | 0 | 0 | 0 | 0 | 0 |
| Aggregate Systems | 0 | 0 | 0 | 0 | 0 | 0 |
| Chips | 0 | 0 | 0 | 0 | 0 | 0 |
| Revenue | ||||||
| System Royalities (from OEM) | 0 | 0 | 0 | 0 | 0 | 0% |
| Sensor Chips | 0 | 0 | 0 | 0 | 0 | 0% |
| Contract and Grants | 0 | 0 | 300,000 | 0 | 300,000 | 100% |
| Total Revenue | 0 | 0 | 300,000 | 0 | 300,000 | 100% |
| Cost of Sales | ||||||
| Sensor Chips | 0 | 0 | 0 | 0 | 0 | 0% |
| Contract and Grants | 0 | 0 | 0 | 0 | 0 | 0% |
| Gross Margin | 0 | 0 | 300,000 | 0 | 300,000 | 100% |
| Operating Expenses | ||||||
| Royalty to University | 0 | 0 | 0 | 0 | 0 | 0% |
| Research & Development | 30,000 | 30,000 | 52,357 | 169,880 | 282,238 | 94% |
| Sales & Marketing | 0 | 0 | 94,804 | 94,804 | 189,608 | 63% |
| General and Administration | 107,017 | 189,658 | 199,497 | 204,417 | 700,589 | 234% |
| Sponsored Research | 450,000 | 0 | 300,000 | 0 | 750,000 | 250% |
| Total Operating Expenses | 587,017 | 219,658 | 646,659 | 469,101 | 1,922,435 | 641% |
| EBIT | (587,017) | (219,658) | (346,659) | (469,101) | (1,622,435) | -541% |
| Interest Expense | (67) | (128) | (5,713) | (8,161) | (14,069) | -5% |
| EBT | (587,084) | (219,786) | (352,372) | (477,262) | (1,636,504) | -546% |
| Income taxes | 0 | 0 | 0 | 0 | 0 | 0% |
| Net Income (Loss) | (587,084) | (219,786) | (352,372) | (477,262) | (1,636,504) | -546% |
| FYE 6/30/01 | |||||
| 1Q | 2Q | 3Q | 4Q | Total | % |
| 0 | 20 | 30 | |||
| 0 | 20 | 50 | 50 | ||
| 0 | 232 | 820 | 1,052 | ||
| 0 | 0 | 96,000 | 144,000 | 240,000 | 5% |
| 0 | 0 | 58,083 | 205,000 | 263,083 | 5% |
| 1,875,000 | 0 | 2,875,000 | 0 | 4,750,000 | 90% |
| 1,875,000 | 0 | 3,029,083 | 349,000 | 5,253,083 | 100% |
| 0 | 0 | 278,419 | 315,289 | 593,708 | 11% |
| 1,687,500 | 0 | 1,687,500 | 0 | 3,375,000 | 64% |
| 187,500 | 0 | 1,063,164 | 33,711 | 1,284,375 | 24% |
| 0 | 0 | 12,327 | 87,673 | 100,000 | 2% |
| 417,923 | 618,476 | 550,176 | 683,067 | 2,269,642 | 43% |
| 140,155 | 160,155 | 280,802 | 326,767 | 907,879 | 17% |
| 277,129 | 302,044 | 391,361 | 395,259 | 1,365,793 | 26% |
| 450,000 | 0 | 300,000 | 0 | 750,000 | 14% |
| 1,285,208 | 1,080,675 | 1,534,665 | 1,492,767 | 5,393,314 | 103% |
| (1,097,708) | (1,080,675) | (471,501) | (1,459,056) | (4,108,939) | -78% |
| (7,988) | (7,642) | (8,409) | (9,879) | (33,919) | -1% |
| (1,105,695) | (1,088,317) | (479,910) | (1,468,935) | (4,142,858) | -79% |
| 0 | 0 | 0 | 0 | 0 | 00% |
| (1,105,695) | (1,088,317) | (479,910) | (1,468,935) | (4,142,858) | -79% |
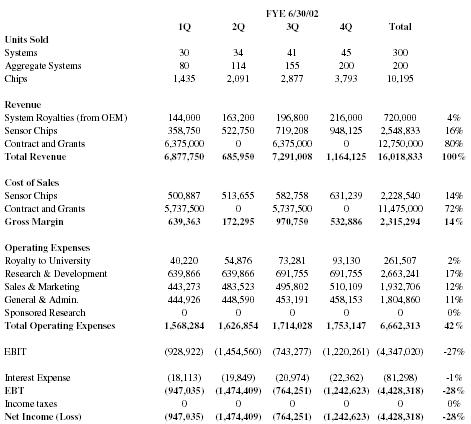
| FYE 6/30/02 | ||||||
| 1Q | 2Q | 3Q | 4Q | Total | ||
| Units Sold | ||||||
| Systems | 30 | 34 | 41 | 45 | 300 | |
| Aggregate Systems | 80 | 114 | 155 | 200 | 200 | |
| Chips | 1,435 | 2,091 | 2,877 | 3,793 | 10,195 | |
| Revenue | ||||||
| System Royalties (from OEM) | 144,000 | 163,200 | 196,800 | 216,000 | 720,000 | 4% |
| Sensor Chips | 358,750 | 522,750 | 719,208 | 948,125 | 2,548,833 | 16% |
| Contract and Grants | 6,375,000 | 0 | 6,375,000 | 0 | 12,750,000 | 80% |
| Total Revenue | 6,877,750 | 685,950 | 7,291,008 | 1,164,125 | 16,018,833 | 100% |
| Cost of Sales | ||||||
| Sensor Chips | 500,887 | 513,655 | 582,758 | 631,239 | 2,228,540 | 14% |
| Contract and Grants | 5,737,500 | 0 | 5,737,500 | 0 | 11,475,000 | 72% |
| Gross Margin | 639,363 | 172,295 | 970,750 | 532,886 | 2,315,294 | 14% |
| Operating Expenses | ||||||
| Royalty to University | 40,220 | 54,876 | 73,281 | 93,130 | 261,507 | 2% |
| Research & Development | 639,866 | 639,866 | 691,755 | 691,755 | 2,663,241 | 17% |
| Sales & Marketing | 443,273 | 483,523 | 495,802 | 510,109 | 1,932,706 | 12% |
| General & Admin. | 444,926 | 448,590 | 453,191 | 458,153 | 1,804,860 | 11% |
| Sponsored Research | 0 | 0 | 0 | 0 | 0 | 0% |
| Total Operating Expenses | 1,568,284 | 1,626,854 | 1,714,028 | 1,753,147 | 6,662,313 | 42% |
| EBIT | (928,922) | (1,454,560) | (743,277) | (1,220,261) | (4,347,020) | -27% |
| Interest Expense | (18,113) | (19,849) | (20,974) | (22,362) | (81,298) | -1% |
| EBT | (947,035) | (1,474,409) | (764,251) | (1,242,623) | (4,428,318) | -28% |
| Income taxes | 0 | 0 | 0 | 0 | 0 | 0% |
| Net Income (Loss) | (947,035) | (1,474,409) | (764,251) | (1,242,623) | (4,428,318) | -28% |
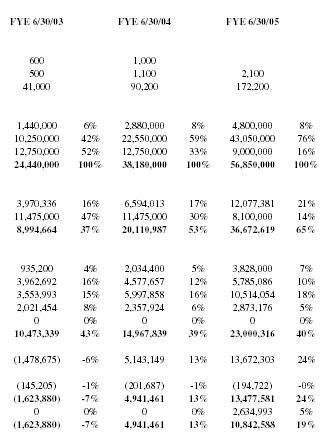
| FYE 6/30/03 | FYE 6/30/04 | FYE 6/30/05 | |||
| 600 | 1,000 | ||||
| 500 | 1,100 | 2,100 | |||
| 41,000 | 90,200 | 172,200 | |||
| 1,440,000 | 6% | 2,880,000 | 8% | 4,800,000 | 8% |
| 10,250,000 | 42% | 22,550,000 | 59% | 43,050,000 | 76% |
| 12,750,000 | 52% | 12,750,000 | 33% | 9,000,000 | 16% |
| 24,440,000 | 100% | 38,180,000 | 100% | 56,850,000 | 100% |
| 3,970,336 | 16% | 6,594,013 | 17% | 12,077,381 | 21% |
| 11,475,000 | 47% | 11,475,000 | 30% | 8,100,000 | 14% |
| 8,994,664 | 37% | 20,110,987 | 53% | 36,672,619 | 65% |
| 935,200 | 4% | 2,034,400 | 5% | 3,828,000 | 7% |
| 3,962,692 | 16% | 4,577,657 | 12% | 5,785,086 | 10% |
| 3,553,993 | 15% | 5,997,858 | 16% | 10,514,054 | 18% |
| 2,021,454 | 8% | 2,357,924 | 6% | 2,873,176 | 5% |
| 0 | 0% | 0 | 0% | 0 | 0% |
| 10,473,339 | 43% | 14,967,839 | 39% | 23,000,316 | 40% |
| (1,478,675) | -6% | 5,143,149 | 13% | 13,672,303 | 24% |
| (145,205) | -1% | (201,687) | -1% | (194,722) | -0% |
| (1,623,880) | -7% | 4,941,461 | 13% | 13,477,581 | 24% |
| 0 | 0% | 0 | 0% | 2,634,993 | 5% |
| (1,623,880) | -7% | 4,941,461 | 13% | 10,842,588 | 19% |
Balance Sheet

| FY 6/30/00 | ||||
| 1Q | 2Q | 3Q | 4Q | |
| ASSETS | ||||
| Current Assets | ||||
| Cash | 784,800 | 473,297 | 8,198,949 | 6,730,576 |
| Net A/R | 0 | 0 | 0 | 0 |
| Inventory | 0 | 0 | 0 | 0 |
| Total Current Assets | 784,800 | 473,297 | 8,198,949 | 6,730,576 |
| Gross Fixed Assets | 10,000 | 20,000 | 70,500 | 1,021,500 |
| Less Acc. Depr. | 567 | 1,700 | 5,356 | 16,833 |
| Net Fixed Assets | 9,433 | 18,300 | 65,144 | 1,004,667 |
| Other Assets | 140,000 | 240,000 | 340,000 | 400,000 |
| TOTAL ASSETS | 934,233 | 731,597 | 8,604,094 | 8,135,242 |
| LIABILITIES | ||||
| Short Term Liabilities | ||||
| A/P | 10,900 | 14,300 | 38,400 | 57,450 |
| Salaries Payable | 5,833 | 15,417 | 25,630 | 38,130 |
| Taxes Payable | 0 | 0 | 0 | 0 |
| Line of Credit | 0 | 0 | 0 | 0 |
| CP- LT Debt | 1,528 | 2,917 | 101,991 | 97,611 |
| Total Short Term Liabilities | 18,261 | 32,633 | 166,021 | 193,191 |
| Long Term Debt | 3,056 | 5,833 | 397,315 | 378,556 |
| TOTAL LIABILITIES | 21,317 | 38,467 | 563,335 | 571,746 |
| Equity | ||||
| Preferred Stock | 1,500,000 | 1,500,000 | 9,200,000 | 9,200,000 |
| Retained Earnings | (587,084) | (806,870) | (1,159,242) | (1,636,504) |
| Total Equity | 912,916 | 693,130 | 8,040,758 | 7,563,496 |
| LIABILITIES & EQUITY | 934,233 | 731,597 | 8,604,094 | 8,135,242 |
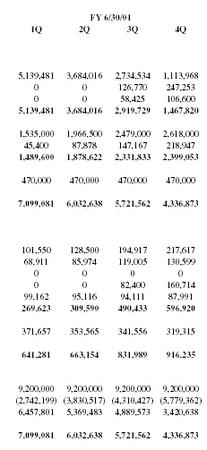
| FY 6/30/01 | |||
| 1Q | 2Q | 3Q | 4Q |
| 5,139,481 | 3,684,016 | 2,734,534 | 1,113,968 |
| 0 | 0 | 126,770 | 247,253 |
| 0 | 0 | 58,425 | 106,600 |
| 5,139,481 | 3,684,016 | 2,919,729 | 1,467,820 |
| 1,535,000 | 1,966,500 | 2,479,000 | 2,618,000 |
| 45,400 | 87,878 | 147,167 | 218,947 |
| 1,489,600 | 1,878,622 | 2,331,833 | 2,399,053 |
| 470,000 | 470,000 | 470,000 | 470,000 |
| 7,099,081 | 6,032,638 | 5,721,562 | 4,336,873 |
| 101,550 | 128,500 | 194,917 | 217,617 |
| 68,911 | 85,974 | 119,005 | 130,599 |
| 0 | 0 | 0 | 0 |
| 0 | 0 | 82,400 | 160,714 |
| 99,162 | 95,116 | 94,111 | 87,991 |
| 269,623 | 309,590 | 490,433 | 596,920 |
| 371,657 | 353,565 | 341,556 | 319,315 |
| 641,281 | 663,154 | 831,989 | 916,235 |
| 9,200,000 | 9,200,000 | 9,200,000 | 9,200,000 |
| (2,742,199) | (3,830,517) | (4,310,427) | (5,779,362) |
| 6,457,801 | 5,369,483 | 4,889,573 | 3,420,638 |
| 7,099,081 | 6,032,638 | 5,721,562 | 4,336,873 |
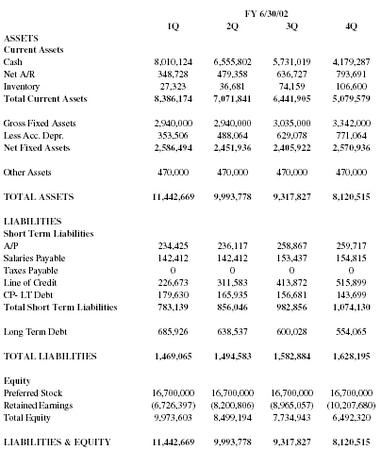
| FY 6/30/02 | ||||
| 1Q | 2Q | 3Q | 4Q | |
| ASSETS | ||||
| Current Assets | ||||
| Cash | 8,010,124 | 6,555,802 | 5,731,019 | 4,179,287 |
| Net A/R | 348,728 | 479,358 | 636,727 | 793,691 |
| Inventory | 27,323 | 36,681 | 74,159 | 106,600 |
| Total Current Assets | 8,386,174 | 7,071,841 | 6,441,905 | 5,079,579 |
| Gross Fixed Assets | 2,940,000 | 2,940,000 | 3,035,000 | 3,342,000 |
| Less Acc. Depr. | 353,506 | 488,064 | 629,078 | 771,064 |
| Net Fixed Assets | 2,586,494 | 2,451,936 | 2,405,922 | 2,570,936 |
| Other Assets | 470,000 | 470,000 | 470,000 | 470,000 |
| TOTAL ASSETS | 11,442,669 | 9,993,778 | 9,317,827 | 8,120,515 |
| LIABILITIES | ||||
| Short Term Liabilities | ||||
| A/P | 234,425 | 236,117 | 258,867 | 259,717 |
| Salaries Payable | 142,412 | 142,412 | 153,437 | 154,815 |
| Taxes Payable | 0 | 0 | 0 | 0 |
| Line of Credit | 226,673 | 311,583 | 413,872 | 515,899 |
| CP- LT Debt | 179,630 | 165,935 | 156,681 | 143,699 |
| Total Short Term Liabilities | 783,139 | 856,046 | 982,856 | 1,074,130 |
| Long Term Debt | 685,926 | 638,537 | 600,028 | 554,065 |
| TOTAL LIABILITIES | 1,469,065 | 1,494,583 | 1,582,884 | 1,628,195 |
| Equity | ||||
| Preferred Stock | 16,700,000 | 16,700,000 | 16,700,000 | 16,700,000 |
| Retained Earnings | (6,726,397) | (8,200,806) | (8,965,057) | (10,207,680) |
| Total Equity | 9,973,603 | 8,499,194 | 7,734,943 | 6,492,320 |
| LIABILITIES & EQUITY | 11,442,669 | 9,993,778 | 9,317,827 | 8,120,515 |
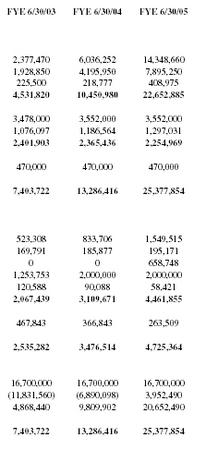
| FYE 6/30/03 | FYE 6/30/04 | FYE 6/30/05 |
| 2,377,470 | 6,036,252 | 14,348,660 |
| 1,928,850 | 4,195,950 | 7,895,250 |
| 225,500 | 218,777 | 408,975 |
| 4,531,820 | 10,450,980 | 22,652,885 |
| 3,478,000 | 3,552,000 | 3,552,000 |
| 1,076,097 | 1,186,564 | 1,297,031 |
| 2,401,903 | 2,365,436 | 2,254,969 |
| 470,000 | 470,000 | 470,000 |
| 7,403,722 | 13,286,416 | 25,377,854 |
| 523,308 | 833,706 | 1,549,515 |
| 169,791 | 185,877 | 195,171 |
| 0 | 0 | 658,748 |
| 1,253,753 | 2,000,000 | 2,000,000 |
| 120,588 | 90,088 | 58,421 |
| 2,067,439 | 3,109,671 | 4,461,855 |
| 467,843 | 366,843 | 263,509 |
| 2,535,282 | 3,476,514 | 4,725,364 |
| 16,700,000 | 16,700,000 | 16,700,000 |
| (11,831,560) | (6,890,098) | 3,952,490 |
| 4,868,440 | 9,809,902 | 20,652,490 |
| 7,403,722 | 13,286,416 | 25,377,854 |
Statment of Cashflow
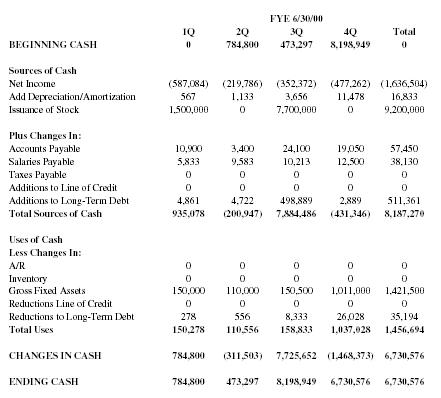
| FYE 6/30/00 | |||||
| 1Q | 2Q | 3Q | 4Q | Total | |
| BEGINNING CASH | 0 | 784,800 | 473,297 | 8,198,949 | 0 |
| Sources of Cash | |||||
| Net Income | (587,084) | (219,786) | (352,372) | (477,262) | (1,636,504) |
| Add Depreciation/Amortization | 567 | 1,133 | 3,656 | 11,478 | 16,833 |
| Issuance of Stock | 1,500,000 | 0 | 7,700,000 | 0 | 9,200,000 |
| Plus Changes In: | |||||
| Accounts Payable | 10,900 | 3,400 | 24,100 | 19,050 | 57,450 |
| Salaries Payable | 5,833 | 9,583 | 10,213 | 12,500 | 38,130 |
| Taxes Payable | 0 | 0 | 0 | 0 | 0 |
| Additions to Line of Credit | 0 | 0 | 0 | 0 | 0 |
| Additions to Long-Term Debt | 4,861 | 4,722 | 498,889 | 2,889 | 511,361 |
| Total Sources of Cash | 935,078 | (200,947) | 7,884,486 | (431,346) | 8,187,270 |
| Uses of Cash | |||||
| Less Changes In: | |||||
| A/R | 0 | 0 | 0 | 0 | 0 |
| Inventory | 0 | 0 | 0 | 0 | 0 |
| Gross Fixed Assets | 150,000 | 110,000 | 150,500 | 1,011,000 | 1,421,500 |
| Reductions Line of Credit | 0 | 0 | 0 | 0 | 0 |
| Reductions to Long-Term Debt | 278 | 556 | 8,333 | 26,028 | 35,194 |
| Total Uses | 150,278 | 110,556 | 158,833 | 1,037,028 | 1,456,694 |
| CHANGES IN CASH | 784,800 | (311,503) | 7,725,652 | (1,468,373) | 6,730,576 |
| ENDING CASH | 784,800 | 473,297 | 8,198,949 | 6,730,576 | 6,730,576 |
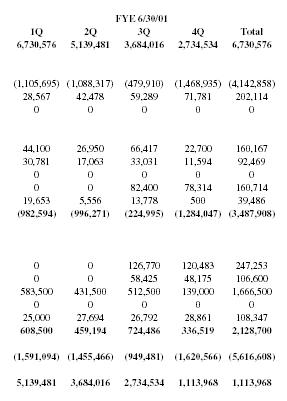
| FYE 6/30/01 | ||||
| 1Q | 2Q | 3Q | 4Q | Total |
| 6,730,576 | 5,139,481 | 3,684,016 | 2,734,534 | 6,730,576 |
| (1,105,695) | (1,088,317) | (479,910) | (1,468,935) | (4,142,858) |
| 28,567 | 42,478 | 59,289 | 71,781 | 202,114 |
| 0 | 0 | 0 | 0 | 0 |
| 44,100 | 26,950 | 66,417 | 22,700 | 160,167 |
| 30,781 | 17,063 | 33,031 | 11,594 | 92,469 |
| 0 | 0 | 0 | 0 | 0 |
| 0 | 0 | 82,400 | 78,314 | 160,714 |
| 19,653 | 5,556 | 13,778 | 500 | 39,486 |
| (982,594) | (996,271) | (224,995) | (1,284,047) | (3,487,908) |
| 0 | 0 | 126,770 | 120,483 | 247,253 |
| 0 | 0 | 58,425 | 48,175 | 106,600 |
| 583,500 | 431,500 | 512,500 | 139,000 | 1,666,500 |
| 0 | 0 | 0 | 0 | 0 |
| 25,000 | 27,694 | 26,792 | 28,861 | 108,347 |
| 608,500 | 459,194 | 724,486 | 336,519 | 2,128,700 |
| (1,591,094) | (1,455,466) | (949,481) | (1,620,566) | (5,616,608) |
| 5,139,481 | 3,684,016 | 2,734,534 | 1,113,968 | 1,113,968 |
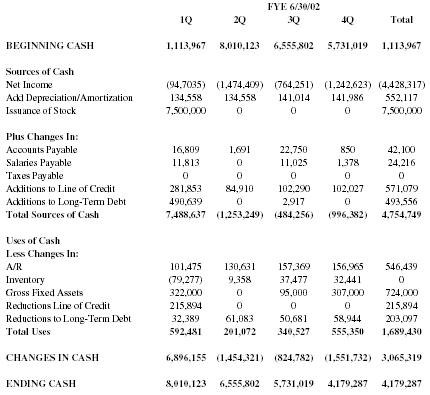
| FYE 6/30/02 | |||||
| 1Q | 2Q | 3Q | 4Q | Total | |
| BEGINNING CASH | 1,113,967 | 8,010,123 | 6,555,802 | 5,731,019 | 1,113,967 |
| Sources of Cash | |||||
| Net Income | (94,7035) | (1,474,409) | (764,251) | (1,242,623) | (4,428,317) |
| Add Depreciation/Amortization | 134,558 | 134,558 | 141,014 | 141,986 | 552,117 |
| Issuance of Stock | 7,500,000 | 0 | 0 | 0 | 7,500,000 |
| Plus Changes In: | |||||
| Accounts Payable | 16,809 | 1,691 | 22,750 | 850 | 42,100 |
| Salaries Payable | 11,813 | 0 | 11,025 | 1,378 | 24,216 |
| Taxes Payable | 0 | 0 | 0 | 0 | 0 |
| Additions to Line of Credit | 281,853 | 84,910 | 102,290 | 102,027 | 571,079 |
| Additions to Long-Term Debt | 490,639 | 0 | 2,917 | 0 | 493,556 |
| Total Sources of Cash | 7,488,637 | (1,253,249) | (484,256) | (996,382) | 4,754,749 |
| Uses of Cash | |||||
| Less Changes In: | |||||
| A/R | 101,475 | 130,631 | 157,369 | 156,965 | 546,439 |
| Inventory | (79,277) | 9,358 | 37,477 | 32,441 | 0 |
| Gross Fixed Assets | 322,000 | 0 | 95,000 | 307,000 | 724,000 |
| Reductions Line of Credit | 215,894 | 0 | 0 | 0 | 215,894 |
| Reductions to Long-Term Debt | 32,389 | 61,083 | 50,681 | 58,944 | 203,097 |
| Total Uses | 592,481 | 201,072 | 340,527 | 555,350 | 1,689,430 |
| CHANGES IN CASH | 6,896,155 | (1,454,321) | (824,782) | (1,551,732) | 3,065,319 |
| ENDING CASH | 8,010,123 | 6,555,802 | 5,731,019 | 4,179,287 | 4,179,287 |
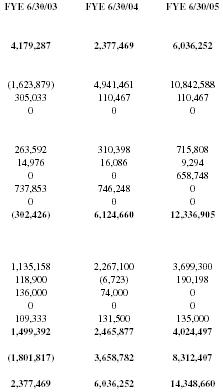
| FYE 6/30/03 | FYE 6/30/04 | FYE 6/30/05 |
| 4,179,287 | 2,377,469 | 6,036,252 |
| (1,623,879) | 4,941,461 | 10,842,588 |
| 305,033 | 110,467 | 110,467 |
| 0 | 0 | 0 |
| 263,592 | 310,398 | 715,808 |
| 14,976 | 16,086 | 9,294 |
| 0 | 0 | 658,748 |
| 737,853 | 746,248 | 0 |
| 0 | 0 | 0 |
| (302,426) | 6,124,660 | 12,336,905 |
| 1,135,158 | 2,267,100 | 3,699,300 |
| 118,900 | (6,723) | 190,198 |
| 136,000 | 74,000 | 0 |
| 0 | 0 | 0 |
| 109,333 | 131,500 | 135,000 |
| 1,499,392 | 2,465,877 | 4,024,497 |
| (1,801,817) | 3,658,782 | 8,312,407 |
| 2,377,469 | 6,036,252 | 14,348,660 |
Comment about this article, ask questions, or add new information about this topic: